- Boateng EB, Ampofo AG. A glimpse into the future: modelling global prevalence of hypertension. BMC Public Health. 2023;23(1):1906. doi:10.1186/s12889-023-16662-z
- Wu X, Sha J, Yin Q, Gu Y, He X. Global burden of hypertensive heart disease and attributable risk factors, 1990–2021: insights from the global burden of disease study 2021. Sci Rep. 2025;15(1):14594. doi:10.1038/s41598-025-99358-1
- Mills KT, Stefanescu A, He J. The global epidemiology of hypertension. Nat Rev Nephrol. 2020;16(4):223–37. doi:10.1038/s41581-019-0244-2
- O’Connell SS, Whelton PK, Li F, Allouch F, Shapiro L, Vandenburg M, et al. Global Hypertension 2000 to 2020: Trends, Disparities, and Progress in Awareness, Treatment, and Control. JACC. 2026. doi: https://doi.org/10.1016/j.jacc.2025.12.091
- Geldsetzer P, Manne-Goehler J, Marcus ME, Ebert C, Zhumadilov Z, Wesseh CS, et al. The state of hypertension care in 44 low-income and middle-income countries: a cross-sectional study of nationally representative individual-level data from 1·1 million adults. The Lancet. 2019 Aug 24;394(10199):652–62. doi:10.1016/S0140-6736(19)30955-9
- World Health Organization. Global report on hypertension: the race against a silent killer. World Health Organization; 2023.
- Burnier M, Egan BM. Adherence in Hypertension. Circ Res. 2019 Mar 29;124(7):1124–40. doi:10.1161/CIRCRESAHA.118.313220
- Zhou B, Carrillo-Larco RM, Danaei G, Riley LM, Paciorek CJ, Stevens GA, et al. Worldwide trends in hypertension prevalence and progress in treatment and control from 1990 to 2019: a pooled analysis of 1201 population-representative studies with 104 million participants. The Lancet. 2021 Sep 11;398(10304):957–80. doi:10.1016/S0140-6736(21)01330-1
- Whelton PK, Robert MC, S AW, E CD, J CK, Cheryl DH, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults. JACC. 2018 May 15;71(19): e127–248. doi: 10.1016/j.jacc.2017.11.006
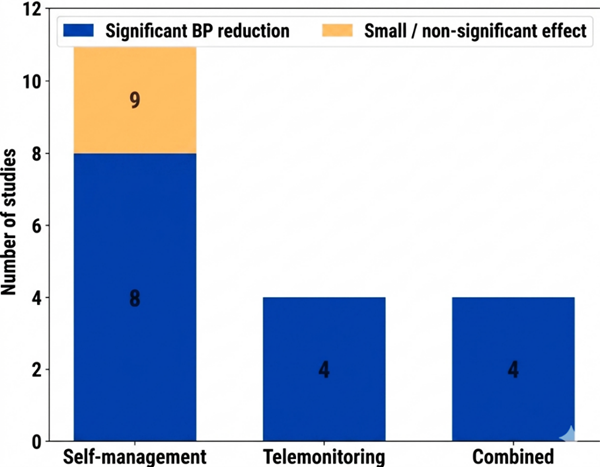
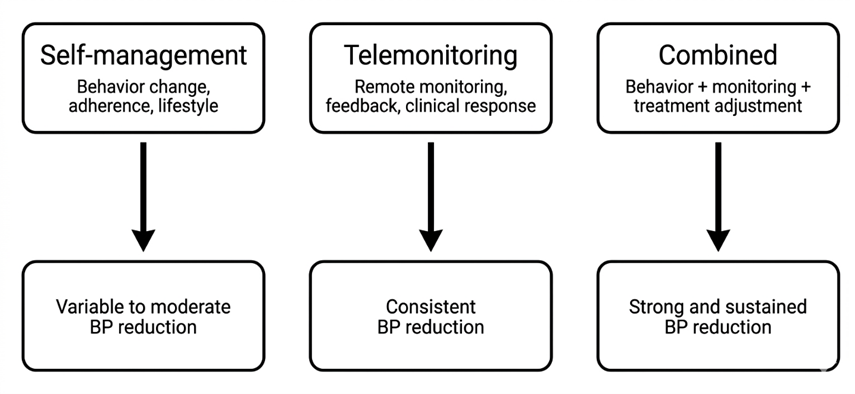
- Boima V, Doku A, Agyekum F, Tuglo LS, Agyemang C. Effectiveness of digital health interventions on blood pressure control, lifestyle behaviours and adherence to medication in patients with hypertension in low-income and middle-income countries: a systematic review and meta-analysis of randomised controlled trials. EClinicalMedicine. 2024 Mar 1;69. doi: 10.1016/j.eclinm.2024.102432
- Ogedegbe G, Schoenthaler A. A Systematic Review of the Effects of Home Blood Pressure Monitoring on Medication Adherence. The Journal of Clinical Hypertension. 2006 Mar 1;8(3):174–80. doi: https://doi.org/10.1111/j.1524-6175.2006.04872.x
- Hwang M, Chang AK. The effect of nurse-led digital health interventions on blood pressure control for people with hypertension: A systematic review and meta-analysis. Journal of Nursing Scholarship. 2023 Sep 1;55(5):1020–35. doi: https://doi.org/10.1111/jnu.12882
- Choudhry NK, Kronish IM, Vongpatanasin W, Ferdinand KC, Pavlik VN, Egan BM, et al. Medication Adherence and Blood Pressure Control: A Scientific Statement from the American Heart Association. Hypertension. 2022 Jan 1;79(1):e1–14. doi:10.1161/HYP.0000000000000203
- Alzahrani SA, Bin Muammar MF, Bin Muammar AF, Alolah A, Almutawa M. The Adoption and Acceptance of mHealth Interventions for Self-Management of Hypertension Among Adult Patients: A Systematic Review. Cureus. 2022 Nov 16. doi:10.7759/cureus.31584 PubMed PMID: 36540533.
- Kassavou A, Wang M, Mirzaei V, Shpendi S, Hasan R. The Association Between Smartphone App–Based Self-monitoring of Hypertension-Related Behaviors and Reductions in High Blood Pressure: Systematic Review and Meta-analysis. JMIR Mhealth Uhealth. 2022;10(7): e34767. doi:10.2196/34767
- Barzegar H, Sodagar S, Seirafi M, Farahbakhsh M, Hashemi T. Blood pressure management protocol based on transtheoretical model effectiveness on self- care: A systematic review. Health Promot Perspect. 2024;14(3):207–20. doi:10.34172/hpp.42814
- Vegesna Ashok, Tran Melody, Angelaccio Michele, Arcona Steve. Remote Patient Monitoring via Non-Invasive Digital Technologies: A Systematic Review. Telemedicine and e-Health. 2017 Jan 1;23(1):3–17. doi:10.1089/tmj.2016.0051
- Masotta V, Dante A, Caponnetto V, Marcotullio A, Ferraiuolo F, Bertocchi L, et al. Telehealth care and remote monitoring strategies in heart failure patients: A systematic review and meta-analysis. Heart & Lung. 2024; 64:149–67. doi: https://doi.org/10.1016/j.hrtlng.2024.01.003
- Zhang Wenhang, Cheng Bo, Zhu Wei, Huang Xiaoxia, Shen Changyin. Effect of Telemedicine on Quality of Care in Patients with Coexisting Hypertension and Diabetes: A Systematic Review and Meta-Analysis. Telemedicine and e-Health. 2021 Jun 1;27(6):603–14. doi:10.1089/tmj.2020.0122
- Parati G, Dolan E, McManus RJ, Omboni S. Home blood pressure telemonitoring in the 21st century. The Journal of Clinical Hypertension. 2018 Jul 1;20(7):1128–32. doi: https://doi.org/10.1111/jch.13305
- Kario K. Management of Hypertension in the Digital Era. Hypertension. 2020 Sep 1;76(3):640–50. doi:10.1161/HYPERTENSIONAHA.120.14742
- Ma Y, Olendzki BC, Wang J, Persuitte GM, Li W, Fang H, et al. Single-Component Versus Multicomponent Dietary Goals for the Metabolic Syndrome. Ann Intern Med. 2015 Feb 17;162(4):248–57. doi:10.7326/M14-0611
- Jaén-Extremera J, Afanador-Restrepo DF, Rivas-Campo Y, Gómez-Rodas A, Aibar-Almazán A, Hita-Contreras F, et al. Effectiveness of Telemedicine for Reducing Cardiovascular Risk: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. MDPI; 2023. doi:10.3390/jcm12030841
- Dones III V, Velasquez AAG, Dacuya MG, Ignacio KET, Cavite ETM, Ibuna II RS, et al. The Effectiveness of Telemedicine in Hypertension Management of Adults in Rural Communities: A Systematic Review and Meta-Analysis. Physiotherapy Research International. 2025 Jan 1;30(1): e70014. doi: https://doi.org/10.1002/pri.70014
- Band R, Bradbury K, Morton K, May C, Michie S, Mair FS, et al. Intervention planning for a digital intervention for self-management of hypertension: a theory-, evidence- and person-based approach. Implementation Science. 2017;12(1):25. doi:10.1186/s13012-017-0553-4
- Band R, Morton K, Stuart B, Raftery J, Bradbury K, Yao GL, et al. Home and Online Management and Evaluation of Blood Pressure (HOME BP) digital intervention for self-management of uncontrolled, essential hypertension: a protocol for the randomised controlled HOME BP trial. BMJ Open. 2016 Nov 1;6(11): e012684. doi:10.1136/bmjopen-2016-012684
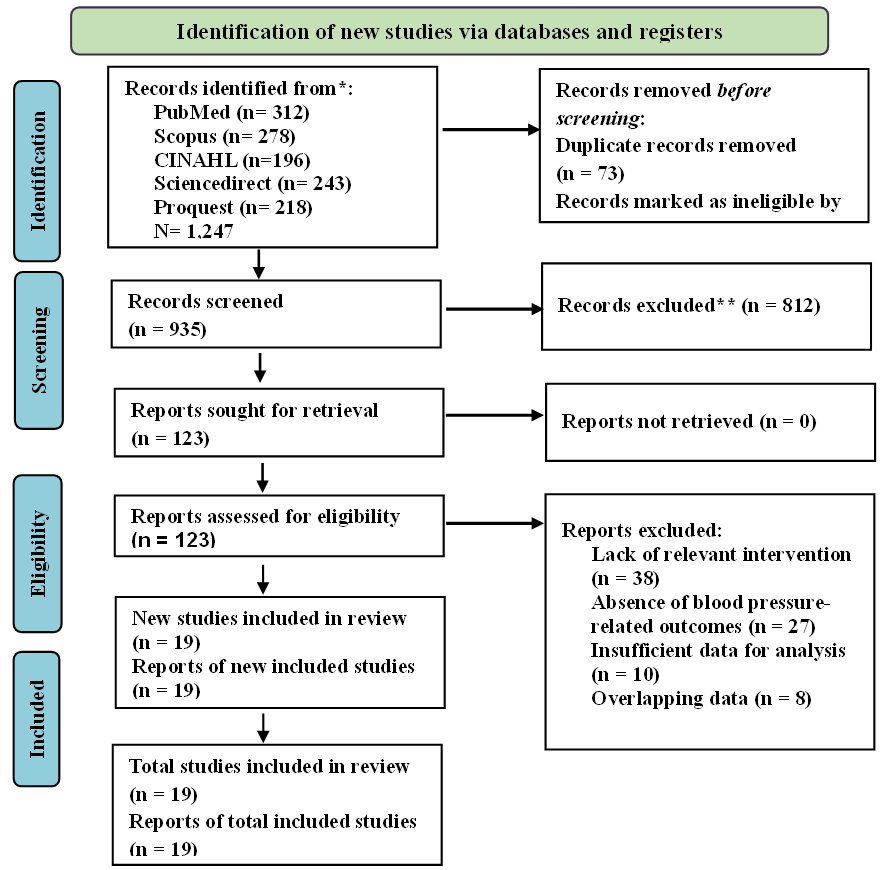
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. The BMJ. 2021 Mar 29;372. doi:10.1136/bmj. n71 PubMed PMID: 33782057.
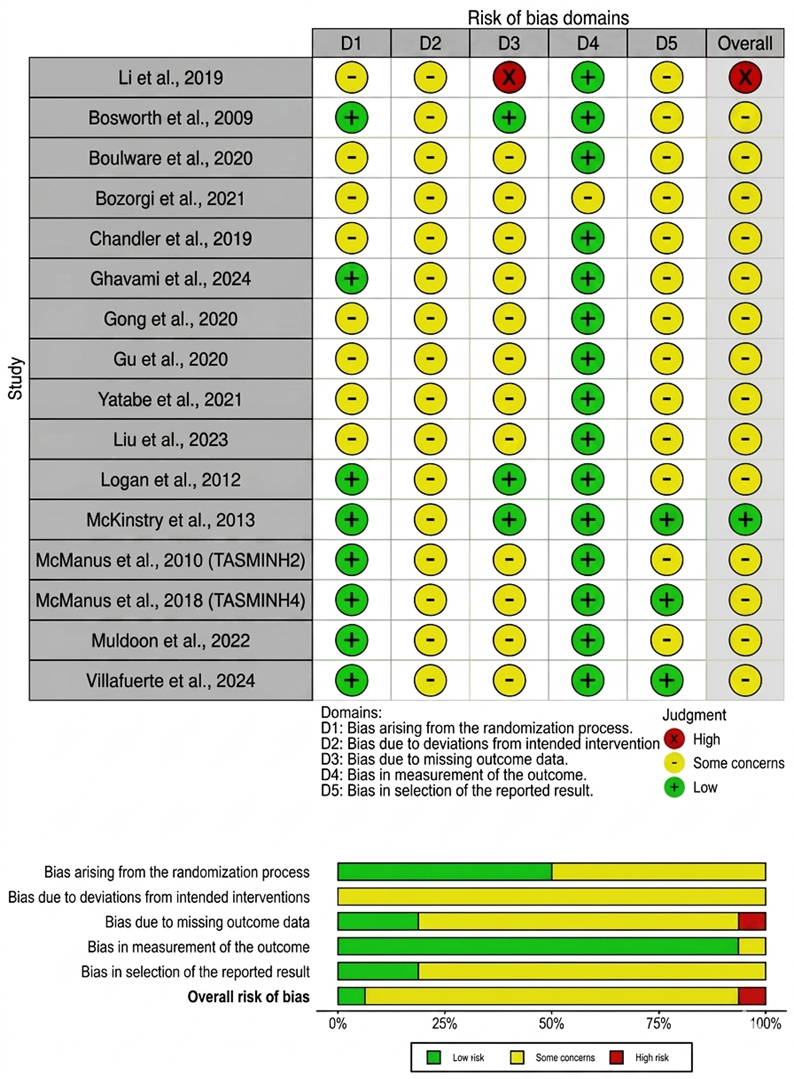
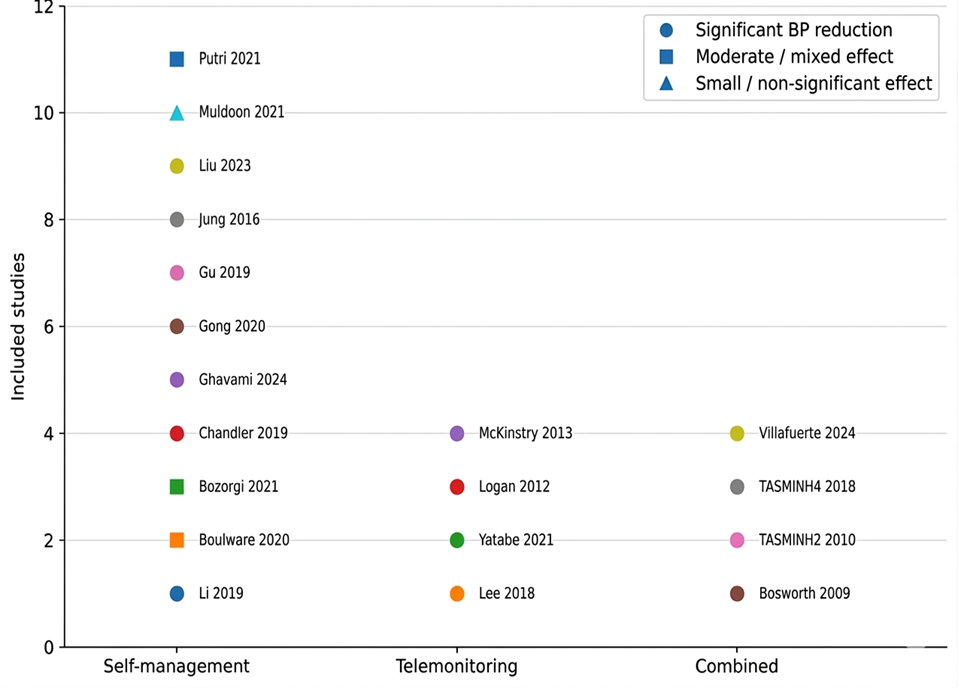
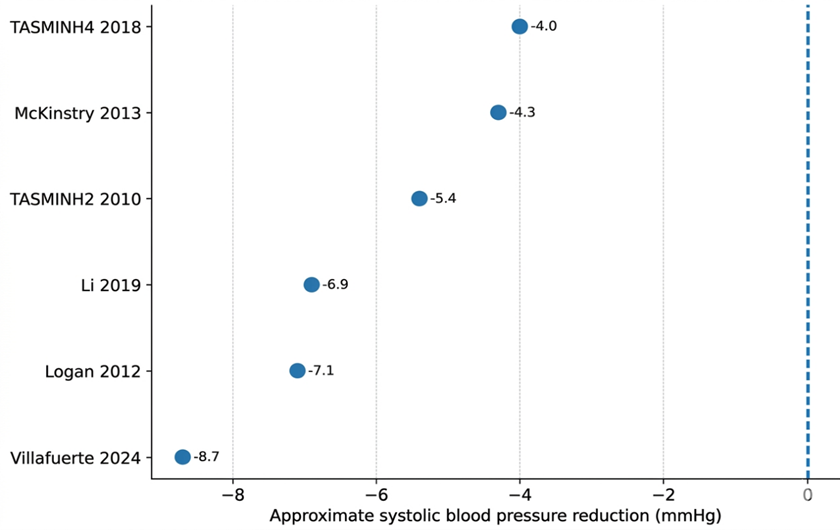
- Li X, Li T, Chen J, Xie Y, An X, Lv Y, et al. A wechat-based self-management intervention for community middle-aged and elderly adults with hypertension in Guangzhou, China: A cluster-randomized controlled trial. Int J Environ Res Public Health. 2019 Nov 1;16(21). doi:10.3390/ijerph16214058 PubMed PMID: 31652688.
- Bosworth HB, Olsen MK, Grubber JM, Neary AM, Orr MM, Powers BJ, et al. Two Self-management Interventions to Improve Hypertension Control A Randomized Trial. Ann Intern Med [Internet]. 2009 Oct 6; 151:687–95. Available from: www.annals.org
- Boulware LE, Ephraim PL, Hill-Briggs F, Roter DL, Bone LR, Wolff JL, et al. Hypertension Self-management in Socially Disadvantaged African Americans: the Achieving Blood Pressure Control Together (ACT) Randomized Comparative Effectiveness Trial. J Gen Intern Med. 2020 Jan 1;35(1):142–52. doi:10.1007/s11606-019-05396-7 PubMed PMID: 31705466.
- Bozorgi A, Hosseini H, Eftekhar H, Majdzadeh R, Yoonessi A, Ramezankhani A, et al. The effect of the mobile “blood pressure management application” on hypertension self-management enhancement: a randomized controlled trial. Trials. 2021 Dec 1;22(1). doi:10.1186/s13063-021-05270-0 PubMed PMID: 34167566.
- Chandler J, Sox L, Kellam K, Feder L, Nemeth L, Treiber F. Impact of a culturally tailored mhealth medication regimen self-management program upon blood pressure among hypertensive hispanic adults. Int J Environ Res Public Health. 2019 Apr 1;16(7). doi:10.3390/ijerph16071226 PubMed PMID: 30959858.
- Ghavami M, Abdshah A, Sadeghian S, Ahmadi A, Jolani MS, Akbarzadeh D, et al. Effectiveness of the Green Heart Smartphone Application as a Self-Management Intervention for Hypertension and Dyslipidemia: A Randomized Clinical Trial. Arch Iran Med. 2024 Jun 1;27(6):313–22. doi:10.34172/aim.28501 PubMed PMID: 38855801.
- Gong K, Yan YL, Li Y, Du J, Wang J, Han Y, et al. Mobile health applications for the management of primary hypertension: A multicenter, randomized, controlled trial. Medicine (United States). 2020 Apr 30;99(16):E19715. doi:10.1097/MD.0000000000019715 PubMed PMID: 32311957.
- Gu Y, Bao X, Wang Y, Meng G, Wu H, Zhang Q, et al. Effects of self-monitoring devices on blood pressure in older adults with hypertension and diabetes: A randomised controlled trial. J Epidemiol Community Health (1978). 2020 Feb 1;74(2):137–43. doi:10.1136/jech-2019-212531 PubMed PMID: 31678965.
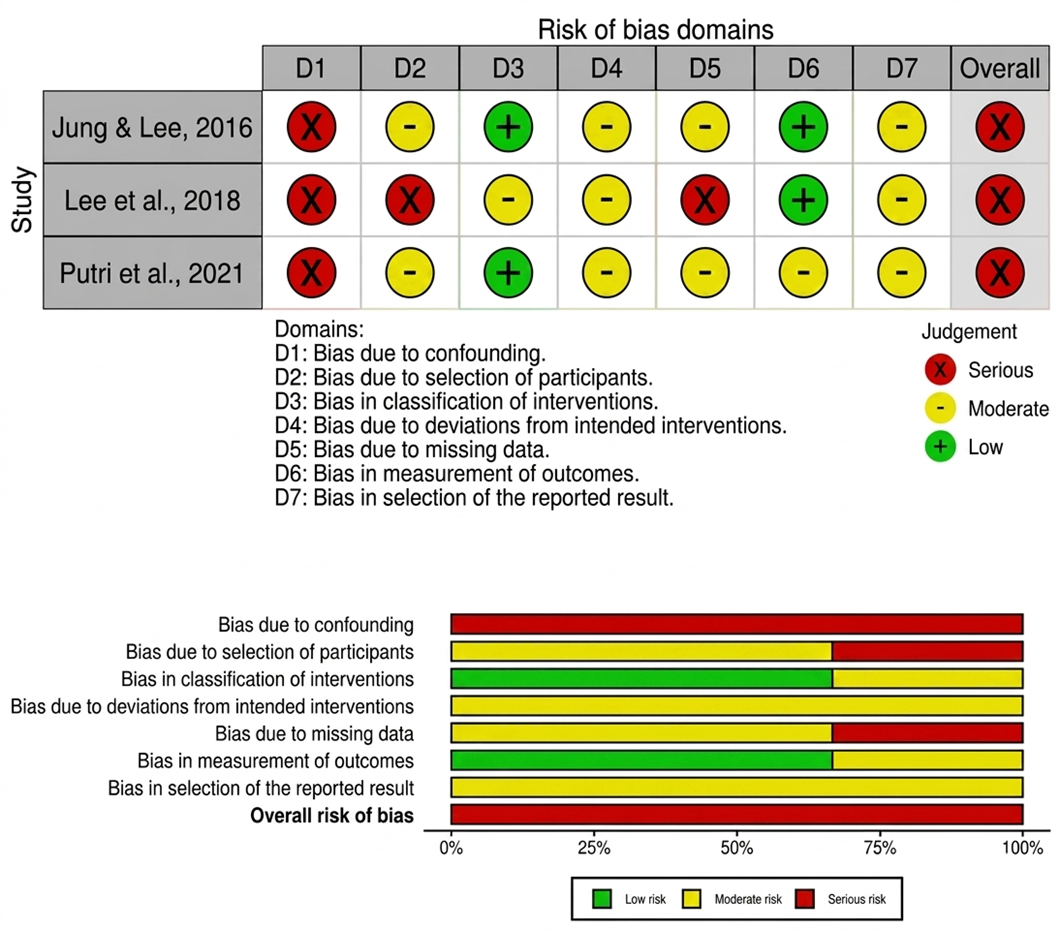
- Jung H, Lee JE. The impact of community-based eHealth self-management intervention among elderly living alone with hypertension. J Telemed Telecare. 2017 Jan 1;23(1):167–73. doi:10.1177/1357633X15621467 PubMed PMID: 26678063.
- Lee HY, Kim JY, Na KY, Park HY, Han J, Pak Y, et al. The role of telehealth counselling with mobile self-monitoring on blood pressure reduction among overseas Koreans with high blood pressure in Vietnam. J Telemed Telecare. 2019 May 1;25(4):241–8. doi:10.1177/1357633X18780559 PubMed PMID: 29933721.
- Yatabe J, Yatabe MS, Okada R, Ichihara A. Efficacy of Telemedicine in Hypertension Care through Home Blood Pressure Monitoring and Videoconferencing: Randomized Controlled Trial. JMIR Cardio. 2021 Jul 1;5(2). doi:10.2196/27347 PubMed PMID: 34321194.
- Liu F, Song T, Yu P, Deng N, Guan Y, Yang Y, et al. Efficacy of an mHealth App to Support Patients’ Self-Management of Hypertension: Randomized Controlled Trial. J Med Internet Res. 2023;25(1). doi:10.2196/43809 PubMed PMID: 38113071.
- Logan AG, Irvine MJ, Mcisaac WJ, Tisler A, Rossos PG, Easty A, et al. Effect of Home Blood Pressure Telemonitoring with Self-Care Support on Uncontrolled Systolic Hypertension in Diabetics. Hypertension. 2012 Jul;60(2012):51–7. doi:10.1161/HYPERTENSIONAHA
- McKinstry B, Hanley J, Wild S, Pagliari C, Paterson M, Lewis S, et al. Telemonitoring based service redesign for the management of uncontrolled hypertension: Multicentre randomised controlled trial. BMJ (Online). 2013 Jun 22;346(7913). doi:10.1136/bmj. f3030 PubMed PMID: 23709583.
- Mcmanus RJ, Mant J, Bray EP, Jones MI, Kaambwa B, Phd B, et al. Telemonitoring and self-management in the control of hypertension (TASMINH2): a randomised controlled trial. Lancet. 2010; 376:163–72. doi:10.1016/S0140
- McManus RJ, Mant J, Franssen M, Nickless A, Schwartz C, Hodgkinson J, et al. Efficacy of self-monitored blood pressure, with or without telemonitoring, for titration of antihypertensive medication (TASMINH4): an unmasked randomised controlled trial. The Lancet. 2018 Mar 10;391(10124):949–59. doi:10.1016/S0140-6736(18)30309-X PubMed PMID: 29499873.
- Muldoon MF, Einhorn J, Yabes JG, Burton D, Irizarry T, Basse J, et al. Randomized feasibility trial of a digital intervention for hypertension self-management. J Hum Hypertens. 2022 Aug 1;36(8):718–25. doi:10.1038/s41371-021-00574-9 PubMed PMID: 34239050.
- Putri SE, Rekawati E, Nurviyandari D, Wati K. Effectiveness of self-management on adherence to self-care and on health status among elderly people with hypertension. Journal of Public Health Research. 2021.
- Villafuerte FU, Cànaves JL, Mantolan AE, Flores PB, Carratalà FR, Hernández AR, et al. Effectiveness of medication self-management, self-monitoring and a lifestyle intervention on hypertension in poorly controlled patients: The MEDICHY randomized trial. Front Cardiovasc Med. 2024 May 21;11. doi:10.3389/fcvm.2024.1355037
- Bludorn J, Railey K. Hypertension Guidelines and Interventions. Primary Care - Clinics in Office Practice. W.B. Saunders; 2024. p. 41–52. doi: 10.1016/j.pop.2023.07.002 PubMed PMID: 38278572.
- Wang Y, Liu Y, Liu L, Hong L, Chen H. Comparative Analysis of Hypertension Guidelines: Unveiling Consensus and Discrepancies in Lifestyle Modifications for Blood Pressure Control. Cardiol Res Pract. 2023 Jan 1;2023(1):5586403. doi: https://doi.org/10.1155/2023/5586403
- Flack JM, Adekola B. Blood pressure and the new ACC/AHA hypertension guidelines. Trends Cardiovasc Med. 2020;30(3):160–4. doi: https://doi.org/10.1016/j.tcm.2019.05.003
- McCarthy CP, Bruno RM, Rahimi K, Touyz RM, McEvoy JW. Global and European landscape of major hypertension guidelines. The Lancet. 2025 Mar 15;405(10482):874–6. doi:10.1016/S0140-6736(24)02795-8
- Tucker KL, Sheppard JP, Stevens R, Bosworth HB, Bove A, Bray EP, et al. Self-monitoring of blood pressure in hypertension: A systematic review and individual patient data meta-analysis. PLoS Med [Internet]. 2017 Sep 19;14(9):e1002389-. Available from: https://doi.org/10.1371/journal.pmed.1002389
- Uhlig K, Patel K, Ip S, Kitsios GD, Balk EM. Self-Measured Blood Pressure Monitoring in the Management of Hypertension. Ann Intern Med. 2013 Aug 6;159(3):185–94. doi:10.7326/0003-4819-159-3-201308060-00008
- Omboni S, Gazzola T, Carabelli G, Parati G. Clinical usefulness and cost effectiveness of home blood pressure telemonitoring: meta-analysis of randomized controlled studies. J Hypertens [Internet]. 2013;31(3). Available from: https://journals.lww.com/jhypertension/fulltext/2013/03000/clinical_usefulness_and_cost_effectiveness_of_home.4.aspx
- Sakima A, Akagi Y, Akasaki Y, Fujii T, Haze T, Kawakami-Mori F, et al. Effectiveness of digital health interventions for telemedicine/telehealth for managing blood pressure in adults: a systematic review and meta-analysis. Hypertension Research. 2025;48(2):478–91. doi:10.1038/s41440-024-01792-7
- Villarino RT, Resti T, Villarino DED. Upgrading Global Hypertension Management through Self-Monitoring and a Culturally Adapted BASNEF Model: A Mixed-method Study in a Resource-Limited Setting. SSRN. 2024 Jul 31. doi:Villarino,RestiTito, UpgradingGlobalHypertensionManagementthroughSelf-MonitoringandaCulturallyAdaptedBASNEFModel: AMixed-MethodStudyinaResource-LimitedSetting(July31,2024). AvailableatSSRN: https://ssrn.com/abstract=4912590orhttp://dx.doi.org/10.2139/ssrn.4912590
- Doogue R, Hayes P, Tucker K, Fahey T, Sheikhi A, Koshiaris C, et al. Self-monitoring for improving control of blood pressure in patients with hypertension. Cochrane Database of Systematic Reviews. 2021 Jan 19;2021(1). doi: 10.1002/14651858.CD010311.pub2
- Mohamadian H, Changizi M, Salehi J, Moridi M. Patient Health Engagement Model (PHE) in Affecting Patient Activation and Self-efficacy Among Cardiac Patients. Jundishapur J Chronic Dis Care. 2024;13(4):1–11. doi: https://doi.org/10.5812/jjcdc-138727
- Chen J, Tian Y, Yin M, Lin W, Tuersun Y, Li L, et al. Relationship between self-efficacy and adherence to self-management and medication among patients with chronic diseases in China: A multicentre cross-sectional study. J Psychosom Res. 2023; 164:111105. doi: https://doi.org/10.1016/j.jpsychores.2022.111105
- Zanatta F, Nissanova E, Świątoniowska-Lonc N, Pierobon A, Callegari G, Olmetti F, et al. Psychosocial Predictors of Self-Efficacy Related to Self-Reported Adherence in Older Chronic Patients Dealing with Hypertension: A European Study. Patient Prefer Adherence. 2020 Sep 25;14(null):1709–18. doi:10.2147/PPA.S258999
- Simmons LA, Wolever RQ, Bechard EM, Snyderman R. Patient engagement as a risk factor in personalized health care: a systematic review of the literature on chronic disease. Genome Med. 2014;6(2):16. doi:10.1186/gm533
- Roncoroni J, Tucker CM, Wall W, Wippold G, Ratchford J. Associations of Health Self-efficacy With Engagement in Health-Promoting Behaviors and Treatment Adherence in Rural Patients. Fam Community Health [Internet]. 2019;42(2). Available from: https://journals.lww.com/familyandcommunityhealth/fulltext/2019/04000/associations_of_health_self_efficacy_with.4.aspx
- Xia T, Zhao F, Nianogo RA. Interventions in hypertension: systematic review and meta-analysis of natural and quasi-experiments. Clin Hypertens. 2022;28(1):13. doi:10.1186/s40885-022-00198-2
- Carey RM, Moran AE, Whelton PK. Treatment of Hypertension: A Review. JAMA. 2022 Nov 8;328(18):1849–61. doi:10.1001/jama.2022.19590
- Kappes M, Espinoza P, Jara V, Hall A. Nurse-led telehealth intervention effectiveness on reducing hypertension: a systematic review. BMC Nurs. 2023;22(1):19. doi:10.1186/s12912-022-01170-z
- Franklin Marika, Willis Karen, Lewis Sophie, Smith Lorraine. Chronic condition self-management is a social practice. Journal of Sociology. 2023 Mar 1;59(1):215–31. doi:10.1177/14407833211038059
- Arbi M, Jamil Q, Sidiq SS, Jamil MN, Iqbal SM. Diabetes, corticosteroids, and mortality outcomes in covid-19-associated rhino-orbital-cerebral mucormycosis. J. Biosci. Public Health. 2026;2(2):139-151. doi:10.5455/JBPH.2026.05
- Meng F, Jiang Y, Yu P, Song Y, Zhou L, Xu Y, et al. Effect of health coaching on blood pressure control and behavioral modification among patients with hypertension: A systematic review and meta-analysis of randomized controlled trials. Int J Nurs Stud. 2023; 138:104406. doi: https://doi.org/10.1016/j.ijnurstu.2022.104406
- Alifariki LO. Various determinants influencing the implementation of occupational health and safety (ohs) in hospitals. J. Biosci. Public Health. 2026;2(1):14-29. doi:10.5455/JBPH.2025.19
- Williams B, Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burnier M, et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology (ESC) and the European Society of Hypertension (ESH). Eur Heart J. 2018 Sep 1;39(33):3021–104. doi:10.1093/eurheartj/ehy339
- K WP, M CR, S AW, E CD, J CK, Cheryl DH, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults. JACC. 2018 May 15;71(19): e127–248. doi: 10.1016/j.jacc.2017.11.006
- Elg M, Kabel D, Gremyr I, Olsson J, Martin J, Smith F. Identification and Categorization of the Distinct Purposes Underpinning the Use of Digital Health Care Self-Monitoring: Qualitative Study of Stakeholders in the Health Care Ecosystem. J Med Internet Res. 2025;27: e58264. doi:10.2196/58264
- Martinengo L, Ha NHL, Tay E, Tong SC, Sevdalis N. Implementation of a Digital Health Intervention (CHAMP) for Self-Monitoring of Hypertension: Protocol for 3 Interlinked Implementation Studies. JMIR Res Protoc. 2025;14: e72942. doi:10.2196/72942
 access
access