J. Biosci. Public Health. 2026; 2(2)
COVID-19-associated mucormycosis is a severe, life-threatening, invasive fungal infection whose incidence was increased substantially in Pakistan during the COVID-19 pandemic. Several studies have described its management, but limited data are available in Pakistan. We assessed the risk factors, treatment modalities, and prognosis of CAM-ROCM to help healthcare providers better manage the disease. A cross-sectional comparative study was conducted to explore demographics, risk factors, clinical manifestations, laboratory parameters, and treatment outcomes of COVID-19-associated rhino-orbital cerebral mucormycosis. A total of 72 patients were included in the study, comprising 41 patients with confirmed COVID-19-associated rhino-orbital cerebral mucormycosis. Most patients were male, aged 40-60 years, and residents of rural areas with diabetes and corticosteroid consumption. Among the stages, rhino-orbital cerebral mucormycosis (41.5%) was the most prevalent, followed by sino-orbital (29.2%), sino-cerebral (17.1%), and rhino-sino mucormycosis (12.2%). Common clinical symptoms included black-brownish nasal discharge, unilateral facial pain, swelling, malaise, fever, eyelid edema, and ophthalmalgia. Most of the patients (82.9%) received amphotericin B deoxycholate. After one week of therapy, blood urea and creatinine levels were elevated, while hypokalemia was observed. A total of 18 patients died, while Hb1Ac, hospital stay duration, steroid therapy, mucormycosis type, and blood urea, creatinine, and potassium levels were significant predictors of survival. Diabetic patients consuming corticosteroids are vulnerable to COVID-19-associated mucormycosis. An uninterrupted supply of liposomal amphotericin B is crucial, and patients receiving therapy should also be observed for the signs of nephrotoxicity.
Mucormycosis is a rare, opportunistic, but deadly, angio-invasive fungal infection caused by Mucorales, a family of Zygomycetes [1]. It generally targets individuals with a suppressed immune system, particularly those suffering from hematological malignancies, solid organ transplants, cancer chemotherapy, immunotherapy, high-dose corticosteroids, uncontrolled diabetes mellitus, and prolonged neutropenia [2]. Mucormycosis is classified by site of infection, most commonly affecting the sinuses and nasal tissues, and can progress to the orbits and brain, known as rhino-orbital cerebral mucormycosis (ROCM). It can also appear in cutaneous, pulmonary, renal, gastrointestinal, and disseminated forms [1]. COVID-19-associated mucormycosis (CAM) is a confirmed infection of mucormycosis during or after SARS-CoV-2 infection [3]. Corticosteroid consumption during COVID-19 and uncontrolled diabetes together constituted an augmented risk factor, which was evidenced by a dramatic increase in the incidence of CAM, with 10% pulmonary and 90% ROCM cases in India [4]. Mucorales spores are airborne and inhaled, where they are deposited in the sinuses or adhered to the nasal epithelium, consequently leading to ROCM as the most common clinical presentation with nasal congestion, brownish or bloody discharge, facial pain, sinus tenderness, nausea, fever, and headache. Nasal and/or palate ulcers appear, which turn black, and the patient may experience facial numbness [1, 2]. ECMM/MSG guidelines recommend first confirming SARS-CoV-2 infection with a positive PCR test, followed by radiographic and histopathologic evidence of mucormycosis. Additionally, imaging techniques, including CT and MRI, should also be performed [5]. Clinical management of mucormycosis is the same in both COVID-19 and non-COVID-19 patients, including risk factor reduction, antifungal drugs, and aggressive surgical debridement [5]. Overall, the prognosis of CAM remains poor, with mortality rates between 25% and 49%, and among survivors, the severe morbidity includes loss of vision. [1, 5] Despite improved understanding of the disease and availability of diagnostic facilities and treatment options, CAM survival rates remain suboptimal.
Pakistan is a developing country with limited health care facilities, mainly concentrated in big cities. It has an enormous burden of infectious diseases with no established surveillance mechanism, including fungal infections [6]. Limited diagnostic facilities, a lack of antimicrobial stewardship, the emergence of resistance, and the suboptimal availability of antifungal drugs, along with insufficient infection control practices, further complicate the situation [7]. Even before the emergence of SARS-CoV-2, the prevalence of mucormycosis (14/100,000; 38% mortality) was relatively high in Pakistan compared to other countries in the region [7, 8].
Despite the high prevalence and mortality rate caused by mucormycosis globally, there are very few reports from Pakistan describing risk factors, diagnosis, treatment, and prognosis of ROCM. We aimed to describe the use of available facilities for diagnosis, management strategies adopted by the healthcare professionals, and adverse drug reactions suffered by the patients in Pakistan during the management of CAM.
2.1. Study design, setting, and data collection
A cross-sectional study was conducted in the ENT ward of BVH, Bahawalpur, Pakistan, from May 2021 to July 2022. BVH is a central tertiary care facility in Southern Punjab, Pakistan, with 1600 beds, providing medical treatment to the local patients and referred patients from other cities of Southern Punjab [9]. BVH is a main health care facility for the population of the Bahawalpur district, which covers 44,588 square kilometers and includes approximately 11.46 million people, of whom 78% reside in rural areas [10-13]. After a thorough literature review by two independent researchers, a new data collection tool was developed. The data collection tool covers sociodemographic information, clinical and laboratory investigations, and patient outcomes. Content and face validity were established through independent assessments by two subject-matter experts. One of the experts had a PhD in pharmacy practice, and the other was an ENT specialist. Clinical data were obtained from medical records and patients, including demographic information, risk factors, clinical, laboratory, and radiological investigations, anti-fungal treatment, surgical intervention, and disease outcomes [12]. Surviving patients were followed up three months post-treatment to assess clinical outcomes.
2.2. Study population and case definition
The criterion for inclusion was i) COVID-19 confirmation with a positive result of RT-PCR from naso/oropharyngeal swabs and ii) histopathological examination with PAS (Periodic Acid-Schiff) stain to confirm mucormycosis. WHO guidelines regarding mild, moderate, and severe SARS-CoV-2 infection were followed [14]. The clinical form, location, and spread of mucormycosis were assessed using CT scans of the orbits, paranasal sinuses, and lungs, and an MRI of the orbits, paranasal sinuses, and brain was performed in some patients as needed. Patients with incomplete medical records (lack of PAS staining or absence of COVID-19 confirmation by RT-PCR) were excluded.
2.3. Ethical considerations
All ethical considerations were observed in this study, in accordance with the principles established by the Declaration of Helsinki and approved by the Pharmacy Research and Ethics Committee (PREC) of the Islamia University of Bahawalpur, Pakistan (142-2021/PREC). Participants who provided consent were informed of the study's objectives, and written permission was obtained from patients to publish their clinical/diagnostic data and images.
2.4. Statistical analysis
Data was analyzed using SPSS (IBM Corp. version 20). Descriptive analysis, such as frequencies and percentages, was used to summarize the data. All analyses met the assumptions of the chi-square test, so it was used to compare treatment outcomes with demographics, risk factors, clinical manifestations, and laboratory diagnoses. A 95% confidence interval was used when applying the chi-square test. The relationship between categories was considered significant if p<0.05.
A total of 72 patients with mucormycosis were admitted to the ENT ward at BVH Bahawalpur. Among these, 41 (57%) patients with confirmed CAM (ROCM) were included in the study (Table 1). All patients with missing medical records (i.e., a lack of PAS staining or a lack of COVID-19 confirmation by RT-PCR) were excluded from the study. Most patients were male (28, 68.3%) rather than female (13, 31.7%). Of the nineteen patients, 46.6% were aged 41-60 years. Thirty-four patients (82.9%) resided in rural areas. CAM was diagnosed 11-20 days after COVID-19 diagnosis in twenty-six patients (63.4%); thirty-three (80.5%) patients consumed corticosteroids, and 40 (97.6%) patients had a documented history of diabetes mellitus. The detailed demographic data of patients are presented in the Table 1.
Table 1. Demographics, risk factors, and comorbidities of patients suffering from ROCM (n=41).
| Demographics and risk factors | Total | Percentage (%) | |
| Gender | Male | 28 | 68.3 |
| Female | 13 | 31.7 | |
| Age (years) | 21-40 | 11 | 26.8 |
| 41-60 | 19 | 46.4 | |
| 61-80 | 11 | 26.8 | |
| Residence | Rural | 34 | 82.9 |
| Urban | 7 | 17.1 | |
| Diagnosis of mucormycosis after COVID-19 (days) | 1-10 | 9 | 22.0 |
| 11-20 | 26 | 63.4 | |
| 21-30 | 6 | 16.6 | |
| Hospital stays (days) | 1-5 | 5 | 12.2 |
| 6-10 | 29 | 70.7 | |
| 11-15 | 4 | 9.8 | |
| >15 | 3 | 7.3 | |
| COVID-19 Severity 1 | Mild | 1 | 2.4 |
| Moderate | 31 | 75.6 | |
| Severe | 9 | 22.0 | |
| Assisted ventilation2 | Yes | 7 | 17.1 |
| No | 34 | 82.9 | |
| Corticosteroid use | Yes | 33 | 80.5 |
| No | 8 | 19.5 | |
| Diabetes mellitus | Yes | 40 | 97.6 |
| No | 1 | 2.4 | |
| Hb1Ac | < 6.5 | 15 | 36.6 |
| ≥ 6.5 | 26 | 63.4 | |
| Diabetic ketoacidosis | Yes | 4 | 9.8 |
| No | 37 | 90.2 | |
| Chronic kidney disease/Renal failure | Yes | 5 | 12.2 |
| No | 36 | 87.8 | |
| Hepatitis B | Positive | 1 | 2.4 |
| Negative | 40 | 97.6 | |
| Hepatitis C | Positive | 4 | 9.8 |
| Negative | 37 | 90.2 | |
| Neutropenia3 | Yes | 1 | 2.4 |
| No | 40 | 97.6 | |
| Anemia4 | Yes | 33 | 80.5 |
| No | 8 | 19.5 | |
1 According to WHO guidelines 8.
2 All types of assisted ventilation, including mechanical, invasive, and non-invasive.
3 Neutrophil count <1000 cells/mm3 at the time of diagnosis.
4 Hb level <13 gm/dl in men and <12 gm/dl in women.
In contrast, the detailed clinical characteristics of patients are presented in Table 2. These patients presented black/brownish nasal discharge (27, 65.9%), unilateral peri-orbital facial pain (27, 65.9%), unilateral facial swelling (33, 80.5%), unilateral facial numbness (24, 58.5%), malaise (32, 78%), fever (27, 65.9%), acute vision loss (14, 34.1%), ophthalmalgia (29, 70.7%), eyelid edema (31, 75.6%), ptosis (20, 48.8%), proptosis (22, 53.7%), and orbital cellulitis (12, 29.3%).
Table 2. Clinical manifestations exhibited by ROCM patients were observed during hospitalization.
| Clinical characteristics | Total | Percentage (%) | |
| Blackish brown nasal discharge | Present | 27 | 65.9 |
| Absent | 14 | 34.1 | |
| Unilateral peri-orbital facial pain | Present | 27 | 65.9 |
| Absent | 14 | 34.1 | |
| Unilateral facial swelling | Present | 33 | 80.5 |
| Absent | 8 | 19.5 | |
| Unilateral facial numbness | Present | 24 | 58.5 |
| Absent | 17 | 41.5 | |
| Malaise | Present | 32 | 78.0 |
| Absent | 9 | 22.0 | |
| Fever | >38 °C | 27 | 65.9 |
| 36.1-37.2 °C | 14 | 34.1 | |
| Acute vision loss | Present | 14 | 34.1 |
| Absent | 27 | 65.9 | |
| Eyelid edema | Present | 31 | 75.6 |
| Absent | 10 | 24.4 | |
| Ptosis | Present | 20 | 48.8 |
| Absent | 21 | 52.2 | |
| Proptosis | Present | 22 | 53.7 |
| Absent | 19 | 46.3 | |
| Right orbital cellulites | Present | 12 | 29.3 |
| Absent | 29 | 70.7 | |
| Ophthalmalgia | Present | 29 | 70.7 |
| Absent | 12 | 29.3 | |
Table 3 demonstrates serial renal function and electrolyte parameters at baseline (Day 0), Day 7, and Day 15 among patients with COVID-19–associated ROCM following amphotericin B therapy. The majority of patients had normal serum urea (78%) and creatinine (87.8%) levels at baseline. Treatment-associated nephrotoxicity was indicated by a significant rise in increased serum urea (>20 mg/dL: 70.8%) and creatinine (>1.2 mg/dL: 73.1%) on Day 7. Even though there was some improvement by Day 15, a significant percentage of patients still had abnormal kidney parameters. In a similar vein, hypokalemia (<3.5 mM/L) rose from 24.4% at baseline to 53.6% on Day 7, with a slight rebound by Day 15. Over the course of the follow-up, serum sodium levels mostly stayed within the normal range. These results emphasize the necessity of careful electrolyte and renal monitoring during amphotericin B treatment.
Table 3. Serial changes in renal function and electrolyte parameters during amphotericin B therapy among patients with COVID-19-associated ROCM.
| Lab parameters | Day 0 (%) | Day 7 (%) | Day 15 (%) | |
| Serum urea | 5-20 mg/dL | 32 (78) | 12 (29.2) | 23 (56.1) |
| >20 mg/dL | 9 (22) | 29 (70.8) | 18 (43.9) | |
| Serum creatinine | 0.7-1.2 mg/dL | 36 (87.8) | 11 (26.9) | 26 (63.4) |
| >1.2 mg/dL | 5 (12.2) | 30 (73.1) | 15 (36.8) | |
| Serum K+ levels | 3.5-5 mM/L | 31 (75.6) | 19 (46.4) | 25 (61) |
| <3.5 mM/L | 10 (24.4) | 22 (53.6) | 16 (39) | |
| Serum Na+ levels | 135-138 mM/L | 39 (95.1) | 36 (87.8) | 40 (97.6) |
| <135 mM/L | 2 (4.9) | 5 (12.2) | 1 (2.4) | |
Clinical eye features of a selected ROCM patient are depicted in Figure 1(a). Histopathological examination with H and E staining was performed for all patients. Histological features of a selected ROCM patient are presented in Figure 1(b). CT scans were performed in 36 patients and MRI scans in 23. Among these, 18 patients had both a CT scan and an MRI. An MRI of a selected CAM patient is presented in Figure 1(c and d).
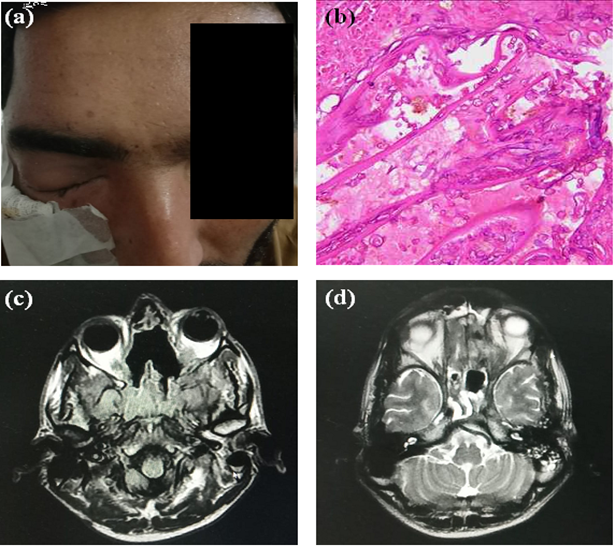
Figure 1. Clinical and diagnostic features of one ROCM patient. (a) Swollen left eye (identity of patient masked); (b) Histological section of respiratory mucosa with underlying necrotic tissue. Many fungal spores and broad, aseptate hyphae are visible. Tissue and vascular invasion are also visible; (c and d) MRI of the brain and orbit with axial T2 FLAIR and FSE view, respectively. Abnormal signal intensity with mucosal thickening was observed in the maxillary, ethmoid, and sphenoid sinuses. Proptosis was noted in the left eye, with abnormal retro-orbital fat and an orbital apex abnormality. The left optic nerve appears swollen, while no extension was observed in the brain tissues.
In Table 4, a total of 41 patients with COVID-19-associated ROCM were included in the analysis. Based on radiological findings (CT/MRI), histopathological confirmation, and intraoperative endoscopic assessment, five patients (12.2%) were diagnosed with rhino-sino mucormycosis, twelve (29.2%) with rhino-orbital mucormycosis, and seven (17.1%) with sino-cerebral mucormycosis, while generalized ROCM was identified in 17 patients (41.5%). According to previously defined severity criteria [15], 21 patients (51.2%) were categorized as moderate and 20 (48.8%) as very severe. Immunohistopathological examination showed that Rhizopus species were the predominant pathogen, identified in 33 patients (80.5%), whereas Mucor species were detected in 8 patients (19.5%).
All patients received systemic antifungal therapy with amphotericin B (5 mg/kg/day). Conventional amphotericin B deoxycholate was administered to 34 patients, whereas 7 patients received liposomal amphotericin B. Among these, two patients additionally received posaconazole oral suspension (800 mg/day in divided doses for four weeks) as combination antifungal therapy. Surgical debridement of necrotic tissues involving the sinuses, palate, and adjacent structures was performed in all patients, with repeated procedures in selected cases. Orbital exenteration was required in six patients due to extensive orbital involvement. Most patients had a documented history of corticosteroid exposure during COVID-19 treatment. In addition, broad-spectrum antibiotics including penicillin, cephalosporins, macrolides, and fluoroquinolones were administered to prevent secondary bacterial infections [16]. To reduce amphotericin B–associated nephrotoxicity, pre-infusion hydration with normal saline was provided according to body weight. Antifungal therapy was temporarily interrupted when laboratory indicators of nephrotoxicity developed, including reduced creatinine clearance, elevated blood urea levels, hypokalemia, or hypomagnesemia, and resumed once renal parameters normalized.
During hospitalization, 18 of the 41 patients (43.9%) died due to complications related to ROCM (Table 4). Mortality did not significantly differ by gender (p = 0.44) or age group (p = 0.22). However, several clinical factors showed significant associations with survival outcomes. Patients with poor glycemic control (HbA1c ≥6.5%) had significantly higher mortality compared with those with HbA1c <6.5% (61.5% vs. 13.3%; p = 0.003). Similarly, hospital stay duration (p = 0.03) and prior corticosteroid use (p = 0.04) were significantly associated with mortality. Disease severity also demonstrated a strong relationship with survival, with patients presenting severe–very severe mucormycosis experiencing substantially higher mortality (70%) compared with those with moderate disease (19%) (p = 0.001). Furthermore, the clinical stage of mucormycosis was significantly associated with mortality (p = 0.04), with the highest mortality observed among patients with generalized ROCM (64.7%) and sino-cerebral involvement (59.1%), whereas patients with rhino-sino and rhino-orbital disease showed comparatively better survival outcomes.
Table 4. Association of sociodemographic characteristics and clinical risk factors with in-hospital mortality among patients with COVID-19-associated ROCM.
| Demographics and risk factors | Survived n (%) | Not survived n (%) | p-value | |
| Gender | Male | 15 (53.6) | 13 (46.4) | 0.44 |
| Female | 8 (61.5) | 5 (38.5) | ||
| Age (Years) | 21-40 | 8 (72.7) | 3 (27.3) | 0.22 |
| 41-60 | 8 (42.1) | 11 (57.9) | ||
| 61-80 | 7 (63.6) | 4 (36.4) | ||
| Hb1AC value | <6.5% | 13 (86.7) | 2 (13.3) | 0.003 |
| ≥6.5% | 10 (38.5) | 16 (61.5) | ||
| Hospital stays (days) | 1-10 | 18 (53) | 16 (47) | 0.03 |
| >10 | 5 (71) | 2 (29) | ||
| Use of corticosteroid (Dexamethasone/ methylprednisolone) | Yes | 16 (48.5) | 17 (51.5) | 0.04 |
| No | 7 (87.5) | 1 (12.5) | ||
Severity of Mucormycosis | Moderate | 17 (81.0) | 4 (19.0) | 0.001 |
| Severe-very severe | 6 (30.0) | 14 (70) | ||
Stage of mucormycosis | Rhino-sino mucormycosis | 4 (80.0) | 1 (20.0) | 0.04 |
| Sino-orbital mucormycosis | 10 (83.3) | 2 (16.7) | ||
| Sino-cerebral mucormycosis | 3 (42.9) | 4 (59.1) | ||
| Generalized ROCM | 6 (35.3) | 11 (64.7) | ||
The association between clinical manifestations and serial laboratory parameters with in-hospital survival among patients with COVID-19-associated ROCM is presented in Table 5. Among the evaluated clinical symptoms, anemia and fever showed significant associations with mortality. Patients with anemia had a significantly higher mortality rate compared with those without anemia (51.5% vs. 12.5%, p = 0.04). Similarly, patients presenting with high fever (>38 °C) had a markedly higher proportion of deaths than those with normal body temperature (59.2% vs. 14.2%, p = 0.008). In contrast, other clinical manifestations including black-brown nasal discharge, unilateral facial pain, and acute vision loss—were not significantly associated with survival outcomes.
Serial monitoring of laboratory parameters revealed significant differences between survivors and non-survivors during the course of hospitalization. While baseline blood urea and serum creatinine levels (Day-0) were not significantly associated with survival, elevated blood urea levels on Day-7 and Day-15 were significantly associated with increased mortality (p = 0.02 and p = 0.002, respectively). Similarly, elevated serum creatinine levels on Day-7 and Day-15 showed strong associations with mortality (p = 0.007 and p = 0.001, respectively).
Table 5. Comparison of clinical manifestations and serial laboratory parameters between survivors and non-survivors with COVID-19-associated ROCM.
Clinical and lab parameters | Survived n (%) | Not-survived n (%) | p-value | |
| Black brown nasal discharge | Present | 15 (55.6) | 12 (49.4) | 0.59 |
| Absent | 8 (57.1) | 6 (42.9) | ||
| Unilateral facial pain | Present | 16 (59.3) | 11 (40.7) | 0.40 |
| Absent | 7 (50) | 7 (50) | ||
| Acute vision loss | Present | 6 (42.8) | 8 (57.2) | 0.18 |
| Absent | 17 (62.9) | 10 (37.1) | ||
| Anemia1 | Present | 16 (48.5) | 17 (51.5) | 0.04 |
| Absent | 7 (87.5) | 1 (12.5) | ||
| Fever | >38 °C | 11 (40.8) | 16 (59.2) | 0.008 |
| 37.3-38 °C | 0 (0) | 0 (0) | ||
| 36.1-37.2 °C | 12 (85.7) | 2 (14.2) | ||
| Blood urea (mg/dL) | ||||
| Day-0 | 5-20 | 16 (50) | 16 (50) | 0.13 |
| >20 | 7 (77.8) | 2 (22.2) | ||
| Day-7 | 5-20 | 10 (83.3) | 2 (16.7) | 0.02 |
| >20 | 13 (44.2) | 16 (55.2) | ||
| Day-15 | 5-20 | 18 (78.3) | 5 (21.7) | 0.002 |
| >20 | 5 (27.8) | 13 (72.2) | ||
| Serum creatinine (mg/dL) | ||||
| Day-0 | 0.7-1.2 | 20 (55.6) | 16 (44.4) | 0.59 |
| >1.2 | 3 (60.0) | 2 (40.0) | ||
| Day-7 | 0.7-1.2 | 10 (90.9) | 1 (9.1) | 0.007 |
| >1.2 | 13 (43.3) | 17 (56.7) | ||
| Day-15 | 0.7-1.2 | 20 (76.9) | 6 (23.1) | 0.001 |
| >1.2 | 3 (20.0) | 12 (80.0) | ||
| Serum K+ levels (mM/L) | ||||
| Day-0 | 3.5-5 | 16 (51.6) | 15 (48.4) | 0.25 |
| <3.5 | 7 (70.0) | 3 (30.0) | ||
| Day-7 | 3.5-5 | 14 (68.0) | 5 (32.0) | 0.03 |
| <3.5 | 9 (40.9) | 13 (59.1) | ||
| Day-15 | 3.5-5 | 17 (68.0) | 8 (32.0) | 0.04 |
| <3.5 | 6 (37.5) | 10 (62.5) | ||
| Serum Na+ levels (mM/L) | ||||
| Day-0 | 135-138 | 23 (59.0) | 16 (41.0) | 0.18 |
| <135 | 0 (0.0) | 2 (100) | ||
| Day-7 | 135-138 | 21 (58.3) | 15 (41.7) | 0.38 |
| <135 | 2 (40.0) | 3 (60.0) | ||
| Day-15 | 135-138 | 23 (57.5) | 17 (42.5) | 0.43 |
| <135 L | 0 (0.0) | 1 (100) | ||
1Hb level <13 g/dL in men and <12 g/dL in women.
Electrolyte imbalance also demonstrated a significant relationship with survival. Hypokalemia (serum K⁺ <3.5 mM/L) on Day-7 and Day-15 was significantly more common among non-survivors compared with survivors (p = 0.03 and p = 0.04, respectively). In contrast, serum sodium levels did not show a statistically significant association with survival at any time point during hospitalization. Overall, these findings indicate that anemia, high fever, and the development of renal dysfunction and electrolyte imbalance during treatment are significantly associated with mortality in patients with COVID-19-associated ROCM.
COVID-19-associated invasive fungal infections caused by Mucorales, Aspergillus, and Candida species constituted a serious complication in a significant number of critically ill patients. CAM was highlighted during the second wave of COVID-19 in early 2021, when more than 47,500 cases were reported in India [17]. The principal modifiable risk factors were the use of systemic corticosteroids and poorly controlled diabetes mellitus [17]. Pakistan, a neighboring country, also saw an increase in CAM incidence, which largely went unreported due to a lack of an effective surveillance system. We identified 72 mucormycosis patients, of whom 41 (56.9%) were confirmed ROCM patients, over 15 months of study at a single tertiary care unit, BVH Bahawalpur. Another Pakistani study found that 51.1% of mucormycosis cases were COVID-19-associated [6]. Among 41 ROCM patients, 80.5% had consumed corticosteroids for COVID-19 management, while 97.6% were known diabetics at the time of diagnosis. These findings indicate that diabetic COVID-19 patients who have consumed systemic corticosteroids have a higher prevalence of CAM infection. Corticosteroid consumption can deregulate glucose metabolism, augment immunosuppression, and predispose such patients to CAM [18]. The WHO recommends corticosteroid therapy, including dexamethasone and methylprednisolone, for the treatment of patients with moderate to severe COVID-19. Glucocorticoids can disturb glucose levels even in healthy people, causing glucocorticoid-induced hyperglycemia [1,18]. This augments the risk of developing CAM in diabetic COVID-19 patients who are also receiving corticosteroid therapy [19]. According to the International Diabetes Federation, in 2022, the prevalence of diabetes in Pakistan is 26.7%, ranking it 3rd after China and India [20]. Out of all diabetes patients, males have a higher prevalence of diabetes compared to females (51.17% vs. 48.83%) [21]. This was also reflected in our study, where the majority of ROCM patients were male (68.3%) and lived in rural areas (82.9%).
All patients received amphotericin B (5 mg/kg/day), with 83% receiving amphotericin B deoxycholate and 12.1% receiving liposomal amphotericin B; 4.87% received combination therapy with liposomal amphotericin B and posaconazole (800 mg/day; p.o.). ECMM/MSG guidelines recommended liposomal amphotericin B (5-10 mg/kg/day) as a first-line therapy preferred over amphotericin B deoxycholate due to adverse drug reactions in the therapy period required for CAM [5]. These guidelines also marginally recommend posaconazole oral suspension due to concerns about bioavailability. Regarding the combination therapy of polyenes and azoles, limited data exist on efficacy and potential benefits, so it is marginally recommended [5]. Pakistan is a developing country that imports most active pharmaceutical ingredients. Amphotericin B was unregistered in Pakistan until 2005, and despite the current registration of a colloidal amphotericin B preparation, its availability remains limited [22]. Thus, colloidal and liposomal amphotericin B usually remain an unregistered or unavailable drug in Pakistan. The surge in CAM cases in Pakistan created a severe local shortage, leading to unjustified price hikes. Patients had no option but to import liposomal amphotericin B from neighboring India, which required multiple regulatory approvals and was costly. Since most patients were from rural areas and unable to afford liposomal amphotericin B, it was also difficult to procure, so amphotericin B deoxycholate was used. Serum creatinine and blood urea levels were increased, while hypokalemia was reported in most patients after the first week of therapy, predisposing them to nephrotoxicity. This led to a pause in amphotericin B administration for patients at risk of nephrotoxicity until their renal parameters were normal and electrolyte levels were restored. Surgical debridement was performed in all patients in the study, and 14.6% underwent one-eye exenteration to prevent spread to the cerebral tissues. All patients also received antibiotics along with antifungal therapy. The trend of broad-spectrum antibiotic administration in CAM patients was also observed in other studies that documented its misuse [23]. Most patients received corticosteroids (dexamethasone or methylprednisolone), which are immunosuppressive and increase the risk of nosocomial infections [24]. Thus, prophylactic antibiotic therapy is administered to patients.
The comparison of sociodemographic and risk factors among survivors and non-survivors indicates that HbA1c levels are significantly associated with mortality risk. Similarly, the consumption of systemic corticosteroids was also linked to increased mortality. Other studies also explore that CAM patients who were administered corticosteroids during COVID-19 infection had a 4.19 times greater risk of death as compared to patients who did not receive corticosteroids [25]. At the same time, the type of ROCM was also an important indicator of survival outcome, where cerebral involvement increased the mortality rates. ROCM patients suffering from fever have an increased mortality risk, which was also observed in other studies [26]. The association between anemia and mortality requires further investigation, as previous studies have not consistently identified hematological parameters as prognostic factors [27]. Hypokalemia, along with elevated creatinine and blood urea nitrogen levels, was often observed after one week of amphotericin deoxycholate therapy due to drug-induced nephrotoxicity, which necessitated temporary treatment discontinuation. For patients receiving amphotericin B, it is recommended that blood urea, creatinine, sodium, potassium, and fluid balance be monitored regularly and corrected as needed and that the amphotericin B dose be adjusted based on serum creatinine and blood urea levels. Our study describes risk factors, diagnosis, treatment, and prognosis of ROCM. We aimed to describe the use of available facilities for diagnosis, management strategies adopted by the healthcare professionals, hurdles in treatment, and adverse drug reactions suffered by the patients in Pakistan. We were unable to compare the efficacy of available therapies due to the limited patient access and COVID-19 lockdown restrictions, which are limitations of the study.
Our findings confirm that middle-aged diabetic patients who received corticosteroids during COVID-19 treatment represent a high-risk group for CAM-ROCM development. Liposomal amphotericin B is the drug of choice for CAM and should be registered and available in the country to reduce the nephrotoxicity risk in mucormycosis patients. Close monitoring of renal function and electrolytes is essential, particularly when conventional amphotericin B deoxycholate is used given due to its higher nephrotoxicity risk.
We are thankful to the Deanship of the Faculty of Pharmacy, the Islamia University of Bahawalpur, Pakistan, and Dr. Irshad-ul-Haq, Head of the ENT ward, BVH, Bahawalpur, Pakistan, for facilitating the conduct of the research work
No funding source
No potential competing interest was reported by the author(s).
This study was conducted in accordance with established ethical standards for human-subject research in accordance with the principles established by the Declaration of Helsinki and approved by the Pharmacy Research and Ethics Committee (PREC) of the Islamia University of Bahawalpur, Pakistan (142-2021/PREC). No personally identifiable information was collected, and all data were kept confidential and used solely for academic and research purposes.
Muhammad Arbi: Methodology, investigation, formal analysis; QurratulAin Jamil: Conceptualization, supervising, writing original draft, writing review and editing; Muhammad Nauman Jamil: Data curation, validation, resources, data visualization, writing review and editing; Sheikh Safeena Sidiq: Data curation, validation, resources, data visualization, writing review and editing; Shahid Muhammad Iqbal: Conceptualization, supervising, writing original draft, writing review and editing.
Data will be available on request.

This work is licensed under a
Creative Commons Attribution 4.0 International License
.
You are free to share and adapt this material for any purpose, even commercially, as long as you give appropriate credit.