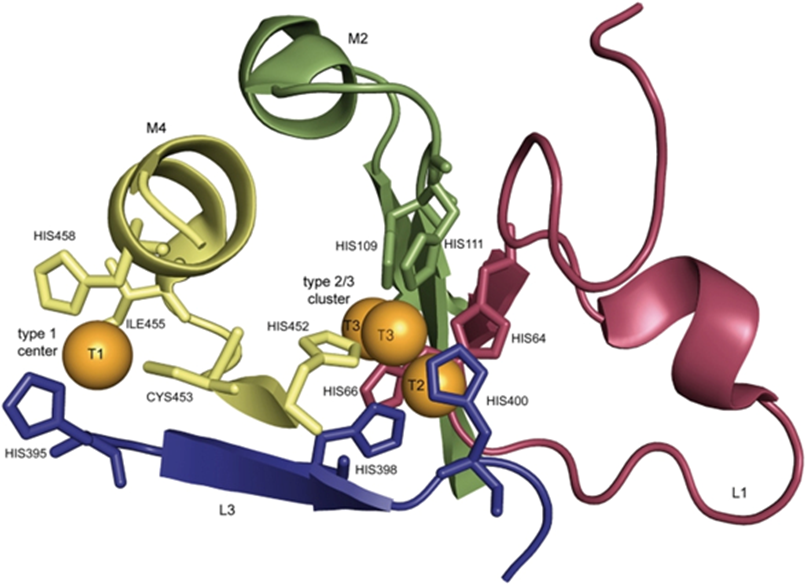
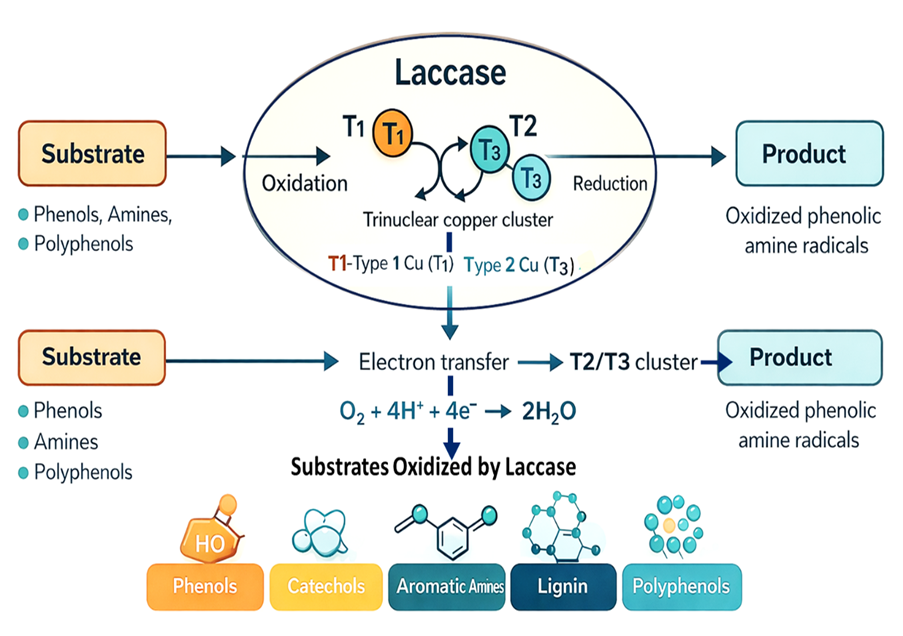
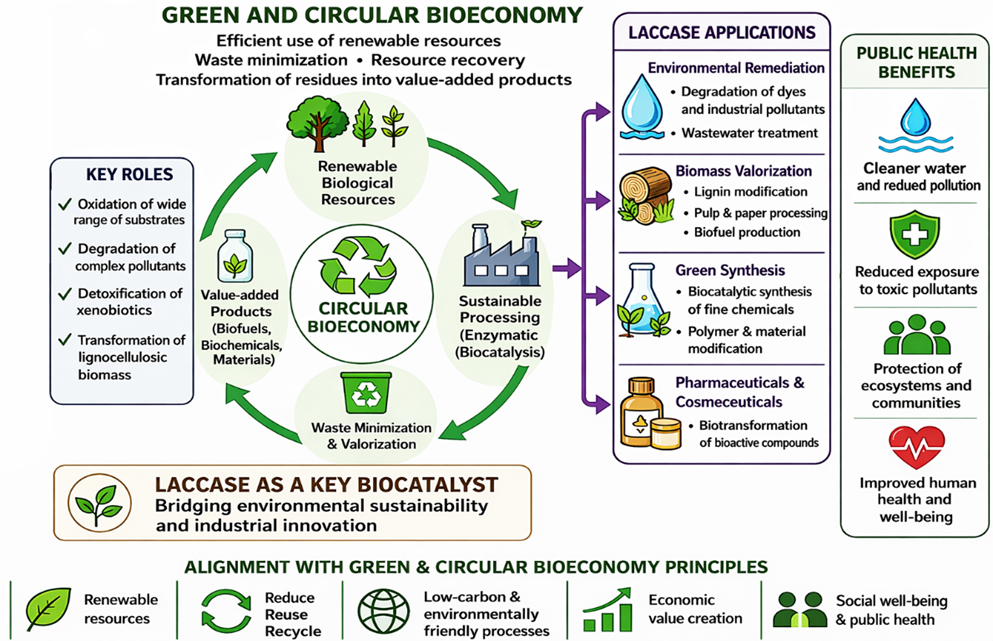
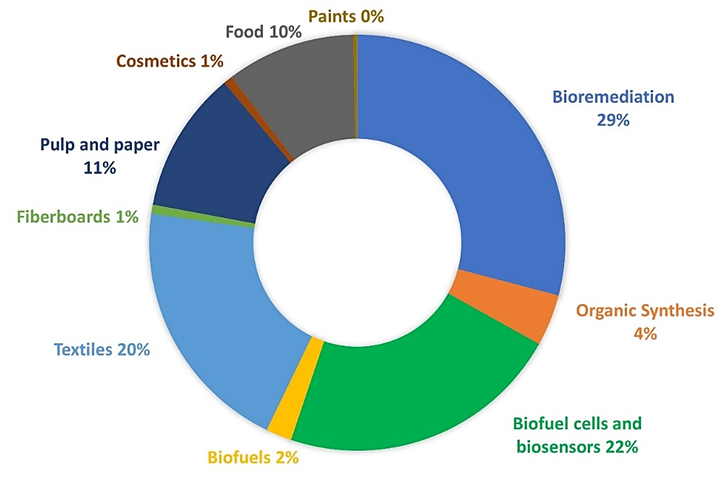
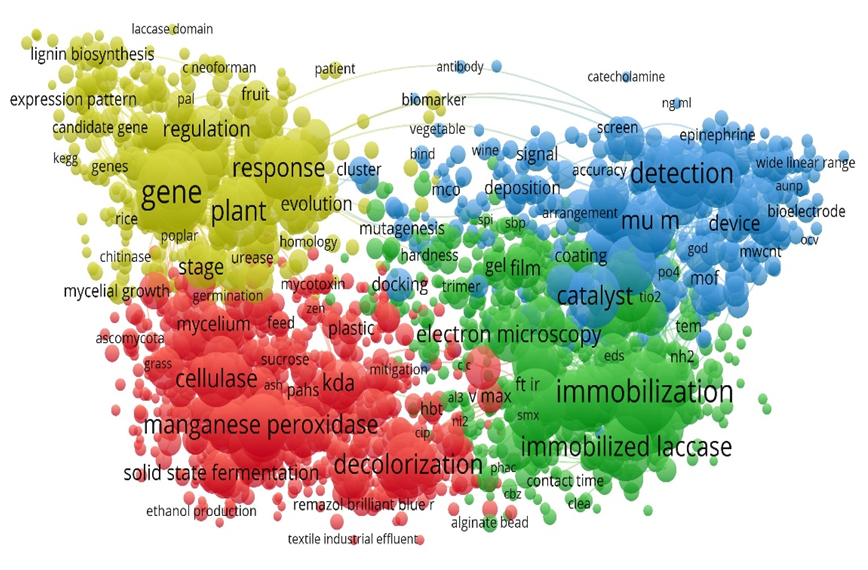
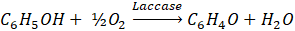- Rochefort D, Kouisni L, Gendron K. Physical immobilization of laccase on an electrode by means of poly(ethyleneimine) microcapsules. J Electroanal Chem. 2008;617(1):53-63. doi: 10.1016/j.jelechem.2008.01.028
- Khatami SH, Vakili O, Khatami M, et al. Laccase: various types and applications. Biotechnol Appl Biochem. 2022;69(6):2658-2672. doi:10.1002/bab.2310
- Paraschiv G, Paraschiv D, Voicu M, et al. Laccases versatile enzymes used to reduce environmental pollution. Energies. 2022; 15:1835. doi:10.3390/en15051835
- Dwivedi UN, Singh P, Pandey VP, Kumar A. Structure–function relationship among bacterial, fungal and plant laccases. J Mol Catal B Enzym. 2011;68(2):117-128. doi: 10.1016/j.molcatb.2010.11.002
- Fernández-Fernández M, Sanromán MÁ, Moldes D. Recent developments and applications of immobilized laccase. Biotechnol Adv. 2013;31(8):1808-1825. doi: 10.1016/j.biotechadv.2013.02.013
- Sharma A, Jain KK, Jain A, et al. Bifunctional in vivo role of laccase exploited in multiple biotechnological applications. Appl Microbiol Biotechnol. 2018;102(24):10327-10343. doi:10.1007/s00253-018-9404-3
- Riva S. Laccases: blue enzymes for green chemistry. Trends Biotechnol. 2006;24(5):219-226. doi: 10.1016/j.tibtech.2006.03.006
- Dubé E, Shareck F, Hurtubise Y, et al. Decolourization of recalcitrant dyes with a laccase from Streptomyces coelicolor under alkaline conditions. J Ind Microbiol Biotechnol. 2008;35(10):1123-1129. doi:10.1007/s10295-008-0393-7
- Ali M, Sreekrishnan TR. Aquatic toxicity from pulp and paper mill effluents: a review. Adv Environ Res. 2001;5(2):175-196. doi:10.1016/S1093-0191(00)00064-7
- Husain Q. Potential applications of the oxidoreductive enzymes in the decolorization and detoxification of textile and other synthetic dyes from polluted water: a review. Crit Rev Biotechnol. 2006;26(4):201-221. doi:10.1080/07388550600969936
- Antošová Z, Sychrová H. Yeast hosts for the production of recombinant laccases: a review. Mol Biotechnol. 2016;58(2):93-116. doi:10.1007/s12033-015-9903-3
- Jaiswal N, Pandey VP, Dwivedi UN. Purification of a thermostable alkaline laccase from papaya (Carica papaya) using affinity chromatography. Int J Biol Macromol. 2015; 72:326-332. doi: 10.1016/j.ijbiomac.2014.08.034
- Solomon EI, Augustine AJ, Yoon J. O₂ reduction to H₂O by multicopper oxidases. Dalton Trans. 2008;(30):3921-3932. doi:10.1039/B800743G
- He F, Machemer-Noonan K, Golfier P, et al. The in vivo impact of MsLAC1, a Miscanthus laccase isoform, on lignification and lignin composition contrasts with its in vitro substrate preference. BMC Plant Biol. 2019; 19:552. doi:10.1186/s12870-019-2137-8
- Shraddha, Shekher R, Sehgal S, et al. Laccase: microbial sources, production, purification, and potential biotechnological applications. Enzyme Res. 2011; 2011:217861. doi:10.4061/2011/217861
- Singh D, Gupta N. Microbial laccase: a robust enzyme and its industrial applications. Biologia. 2020;75(8):1183-1193. doi:10.2478/s11756-020-00492-1
- Kilaru S, Hoegger PJ, Kües U. The laccase multi-gene family in Coprinopsis cinerea has seventeen members that divide into two subfamilies. Curr Genet. 2006;50(1):45-60. doi:10.1007/s00294-006-0074-2
- Desai SS, Nityanand C. Microbial laccases and their applications: a review. Asian J Biotechnol. 2011;3(2):98-124. doi:10.3923/ajbkr.2011.98.124
- Ramayanam BM. Laccase enzyme production from Trametes versicolor: a sustainable approach to treat raw dye bath textile effluent. Essential Chem. 2025;2(1):2527045. doi: 10.1016/j.esschem.2025.2527045
- Oztat K, Yavuz AA, Işçen CF. Optimization studies on laccase activity of Proteus mirabilis isolated from textile sludge. Braz J Microbiol. 2024;55(2):1231-1241. doi:10.1007/s42770-023-01074-4
- Renfeld ZV, Kolesnikova SA, et al. Unusual oligomeric laccase-like oxidases from Curvularia geniculata. Microorganisms. 2023; 11:2698. doi:10.3390/microorganisms11112698
- Cheng Z, Liu Y, Wang X, et al. Cu–Fe₃O₄ nanozyme reverses age-impaired alveolar healing via ROS/RNS scavenging. Langmuir. 2025;41(49):33293-33305. doi: 10.1021/acs.langmuir.5c03021
- Sharma Y, Tiwari A, Singh R, et al. Nanotechnology as a vital science in accelerating biofuel production: boon or bane. Biofuels Bioprod Biorefin. 2023;17(3):616-663. doi:10.1002/bbb.2445
- Akter R, Rahman MS, Islam MS, et al. Hydrobiology of saline agriculture ecosystem in southwest Bangladesh. Hydrobiology. 2023; 2:162-180. doi:10.3390/hydrobiology2010011
- Mate DM, García-Burgos C, García-Ruiz E, et al. Functional expression of a blood tolerant laccase in Pichia pastoris. BMC Biotechnol. 2013; 13:38. doi:10.1186/1472-6750-13-38
- Asaduzzaman D. Cost-benefit analysis of fire safety systems in ready-made garment industry: a case study. Ann Burns Fire Disasters. 2022;35(2):137-142.
- Fang X, Liu Y, Zhang H, et al. Enhanced laccase production by Aspergillus oryzae from lignocellulosic wastes through solid-state fermentation: cytotoxicity, anticancer activity, and 4-chlorophenol degradation. Int J Biol Macromol. 2025; 316:144450. doi: 10.1016/j.ijbiomac.2025.144450
- Hasan S, Rahman MM, Islam MA, et al. Laccase production from local biomass using solid-state fermentation. Fermentation. 2023; 9:179. doi:10.3390/fermentation9020179
- Chaoua S, El Modafar C, Moumni M, et al. Yellow laccase produced by Trametes versicolor K1 on tomato waste: a comparative study with blue laccase. J Biotechnol. 2023; 361:99-109. doi: 10.1016/j.jbiotec.2022.12.010
- Contreras E, Téllez-Jurado A, Rodríguez A, et al. Agro-industrial wastes revalorization as feedstock for lignin-modifying enzyme production by white-rot fungi. Prep Biochem Biotechnol. 2023;53(5):488-499. doi:10.1080/10826068.2022.2098123
- França ED, Silva LF, Souza AF, et al. Valorization of spent coffee grounds as substrate for fungal laccase production and biosorbents for dye decolorization. Fermentation. 2025; 11:396. doi:10.3390/fermentation11070396
- Rezaei S, Shahverdi AR, Faramarzi MA. Isolation, purification, and characterization of a polyextremotolerant laccase from Aquisalibacillus elongatus. Bioresour Technol. 2017; 230:67-75. doi: 10.1016/j.biortech.2017.01.041
- Gupta C, Chauhan SB. Engineered fungal cell factories: pioneering sustainable protein and peptide production. In: Gupta A, ed. Prospects of Fungal Biotechnologies for Livestock. Vol 2. Springer Nature Switzerland; 2026:233-262.
- Pathania AS, Thakur N, Sharma M, et al. Biopharmaceuticals from filamentous fungi. In: Singh RS, Bhari R, eds. Fungal Biotechnology. Academic Press; 2025:253-290.
- Teo HL, Lee CK, Ooi CW, et al. Statistically assisted optimisation for simultaneous enzyme production. Biomass Convers Biorefin. 2025;15(3):3827-3843. doi:10.1007/s13399-023-04025-0
- Mohanty S, Behera SS, Ray RC, et al. Designing a scalable expression platform for recombinant enzyme production in E. coli. Process Biochem. 2025; 150:21-32. doi: 10.1016/j.procbio.2024.11.005
- de Marco A. Recent advances in recombinant production of soluble proteins in E. coli. Microb Cell Fact. 2025; 24:21. doi:10.1186/s12934-025-02234-5
- Mejia-Otalvaro F, López-Gallego F, Rocha-Martin J, et al. Sustainable natural product glycosylation. ChemSusChem. 2025;18(20): e202501094. doi:10.1002/cssc.202501094
- Akher SA, Rahman MS, Islam MA, et al. Harnessing transient expression systems in Nicotiana benthamiana. Int J Mol Sci. 2025; 26:5510. doi:10.3390/ijms26125510
- Ghose S, Banerjee S, Chakraborty R, et al. Pesticides and allergens. In: Pharmacognosy and Phytochemistry. 2025:315-334.
- Ahmad T, Zhang Y, Li X, et al. Processing and bioprocessing developments in alternative proteins. Food Bioprocess Technol. 2025;19(2):54. doi:10.1007/s11947-024-03521-7
- Guo Y, Li X, Zhang H, et al. Development and challenges of forest food resources. Foods. 2025; 14:3503. doi:10.3390/foods14203503
- Günal-Köroğlu D, Demirhan E, Yilmaz MT, et al. Allergenicity of alternative proteins. J Agric Food Chem. 2025;73(13):7522-7546.
- Taheri-Anganeh M, et al. In silico analysis of signal peptides for recombinant enzyme secretion. Mol Biol Res Commun. 2019;8(1):17-26.
- Taheri-Anganeh M, et al. LytU-SH3b fusion protein as enzybiotic against MRSA. Mol Biol Res Commun. 2019;8(4):151-158.
- Taheri-Anganeh M, et al. In silico evaluation of PLAC1-fliC vaccine. Iran Biomed J. 2020;24(3):173-182. doi:10.29252/ibj.24.3.173
- Khatami SH, et al. Signal peptide analysis for recombinant antigen B8/1. Mol Biol Res Commun. 2020;9(1):1-10.
- Taheri-Anganeh M, et al. PRAME+FliCΔD2D3 vaccine design. Iran J Med Sci. 2021;46(1):52-60. (No DOI)
- Dubé E, Shareck F, Hurtubise Y, et al. Cloning and characterization of laccase from Streptomyces coelicolor. Appl Microbiol Biotechnol. 2008;79(4):597-603. doi:10.1007/s00253-008-1454-5
- Bertrand B, Martínez-Morales F, Trejo-Hernández MR. Upgrading laccase production: strategies and challenges. Biotechnol Prog. 2017;33(4):1015-1034. doi:10.1002/btpr.2473
- Nevalainen KMH, Te’o VSJ, Bergquist PL. Heterologous protein expression in filamentous fungi. Trends Biotechnol. 2005;23(9):468-474. doi: 10.1016/j.tibtech.2005.06.002
- Gellissen G, Hollenberg CP. Yeast expression systems: a comparative review. Gene. 1997;190(1):87-97. doi:10.1016/S0378-1119(97)00010-7
- Dittmer NT, Gorman MJ, Kanost MR. Characterization of laccase-2 in Manduca sexta. Insect Biochem Mol Biol. 2009;39(9):596-606. doi: 10.1016/j.ibmb.2009.06.004
- Li L, Steffens JC. Overexpression of polyphenol oxidase enhances disease resistance. Planta. 2002;215(2):239-247. doi:10.1007/s00425-002-0750-2
- Haq I, Saleem A, Chaudhary R, et al. Role of microbial laccases in biomass valorization. Front Bioeng Biotechnol. 2024; 12:1441075. doi:10.3389/fbioe.2024.1441075
- Tiwari A, Singh R, Kumar V, et al. Laccase in biorefinery of lignocellulosic biomass. Appl Sci. 2023;13(8):4673. doi:10.3390/app13084673
- Serbent MP, et al. Immobilizing laccase for organochlorine removal. Biotechnol Bioeng. 2024. doi:10.1002/bit.28591
- Li Z, Wang X, Zhang Y, et al. Co-immobilization of laccase enhances pollutant degradation. Molecules. 2024;29(2):307. doi:10.3390/molecules29020307
- Sharma N, Basera P. Green chemistry strategies in biomass valorization. Front Chem. 2025; 13:1724324. doi:10.3389/fchem.2025.1724324
- Rajendran S, Kalairaj A, Senthilvelan T. Enzymatic decolorization of azo dyes using laccase. Biomass Convers Biorefin. 2025;15(9):13079-13101. doi:10.1007/s13399-023-04321-9
- Mithu, M.M.U., Shormela, S.A., Abdullah, A.T.M. et al. Exploring Heavy Metal Bioaccumulation in Vegetables: Unraveling Environmental Pollutants' Impact on Agricultural Produce and Human Health. Biol Trace Elem Res (2025). https://doi.org/10.1007/s12011-025-04871-z
- Jacob J, Krishnani K. Dehalococcoides-assisted reductive dechlorination. Curr Sci. 2024;127(10). (No DOI)
- Santos MS, et al. Removal of endocrine disruptors using photocatalysis. Int J Environ Res Public Health. 2025;22(3).
- Salehi S, et al. Biocatalysts for oxidation of phenolic pollutants. Sustainability. 2021;13. doi:10.3390/su13158620
- Nunes CS, Malmlöf K. Enzymatic decontamination of pollutants. In: Enzymes in Human and Animal Nutrition. Elsevier; 2018:331-359.
- Padhye LP, Tezel U. Fate of environmental pollutants. Water Environ Res. 2014;86(10):1714-1773. doi:10.2175/106143014X13975035526133
- Lund MN. Reactions of plant polyphenols in foods. Trends Food Sci Technol. 2021; 112:241-251. doi: 10.1016/j.tifs.2021.03.039
- Pillar-Little EA, Zhou R, Guzman MI. Heterogeneous oxidation of catechol. J Phys Chem A. 2015;119(41):10349-10359. doi: 10.1021/acs.jpca.5b07977
- Uyama H, Kobayashi S. Enzymatic synthesis of polymers from polyphenols. In: Kobayashi S, Ritter H, Kaplan D, eds. Enzyme-Catalyzed Synthesis of Polymers. Springer; 2006:51-67. doi:10.1007/3-540-26497-2_3
- Akkineni S, Rawas-Qalaji M, Kouzi SA, Chbib C, Uddin MN. Exploring the Biological Activities of Ionic Liquids and Their Potential to Develop Novel Vaccine Adjuvants. Vaccines. 2025; 13(4):365. https://doi.org/10.3390/vaccines13040365
- Pinar O, Rodríguez-Couto S. Secondary metabolites from white-rot fungi. Front Chem. 2024;12.
- Ren D, et al. Immobilized laccase: environmental applications. Biotechnol Genet Eng Rev. 2020;36(2):81-131. doi:10.1080/02648725.2020.1780141
- Wan Mohtar WHM, et al. Role of fungi in sustainability. Bioengineered. 2022; 13:14903-14935. doi:10.1080/21655979.2022.2091234
- Gholami-Shabani M, Shams-Ghahfarokhi M, Razzaghi-Abyaneh M. Natural product synthesis by fungi. In: Yadav AN, et al., eds. Recent Advancement in White Biotechnology Through Fungi. Springer; 2019:195-228.
- Ferdeș M, et al. Microorganisms and enzymes in biogas pretreatment. Sustainability. 2020;12. doi:10.3390/su12177205
- Sousa AC, Martins LO, Robalo MP. Laccases: versatile biocatalysts for the synthesis of heterocyclic cores. Molecules. 2021; 26:3719. doi:10.3390/molecules26123719
- Nunes CS, Malmlöf K. Enzymatic decontamination of antimicrobials, phenols, heavy metals, pesticides, polycyclic aromatic hydrocarbons, dyes, and animal waste. In: Nunes CS, Kumar V, eds. Enzymes in Human and Animal Nutrition. Academic Press; 2018:331-359.
- Gupta S, Verma A, Sharma P, et al. Microbial laccases: structure, function, and applications. In: Microbial Enzymes. 2025:665-695. (DOI to verify)
- Della Pelle F, Compagnone D. Nanomaterial-based sensing of phenolic compounds and antioxidant capacity in food. Sensors. 2018; 18:462. doi:10.3390/s18020462
- Singh Arora D, Sharma RK. Ligninolytic fungal laccases and their biotechnological applications. Appl Biochem Biotechnol. 2010;160(6):1760-1788. doi:10.1007/s12010-009-8676-y
- Reis GCL, Silva TM, Carvalho AM, et al. Biologically active amines in edible mushrooms. J Food Compos Anal. 2020; 86:103375. doi: 10.1016/j.jfca.2019.103375
- Gangwar R, Singh P, Yadav S, et al. Bioactive proteins from Pleurotus ostreatus: antibiofilm potential. Front Microbiol. 2024;15.
- Al-Maqdi KA, Alshammari AA, et al. Enzyme-mediated wastewater remediation: recent advances. Nanomaterials. 2021; 11:3124. doi:10.3390/nano11113124
- Strong PJ, Claus H. Laccase: past and future in bioremediation. Crit Rev Environ Sci Technol. 2011;41(4):373-434. doi:10.1080/10643380902945706
- Benali J, Ben Atitallah I, Ghariani B, Mechichi T, Hadrich B, Zouari-Mechichi H. Optimized decolorization of azo dyes using laccase mediator system. 3 Biotech. 2024;14(3):93. doi:10.1007/s13205-024-03937-4
- Egbewale SO, Adekunle AA, et al. ABTS-mediated fluoranthene biotransformation by fungal laccases. Int Biodeterior Biodegradation. 2025; 196:105946. doi: 10.1016/j.ibiod.2025.105946
- Viswanath B, Rajesh B, Janardhan A, et al. Fungal laccases in bioremediation. Enzyme Res. 2014; 2014:163242. doi:10.1155/2014/163242
- Rafeeq H, Afsheen N, Rafique S, Arshad A, Intisar M, Hussain A, Bilal M, Iqbal HMN. Genetically engineered microorganisms for environmental remediation. Chemosphere. 2023; 310:136751. doi: 10.1016/j.chemosphere.2022.136751
- Eira A, Gonçalves MBS, Fongang YSF, Domingues C, Jarak I, Mascarenhas-Melo F, Figueiras A. Ganoderma lucidum: therapeutic and nanotechnological advances. Pharmaceutics. 2025;17(4):422. doi:10.3390/pharmaceutics17040422
- Giraldo LDR, Baez PVX, Forero CZJ, Arango WM. Exopolysaccharide production in agaricomycetes. Bio Protoc. 2023;13(19): e4841. doi:10.21769/BioProtoc.4841
- Sekan AS, Myronycheva OS, Karlsson O, Gryganskyi AP, Blume Y. Green potential of Pleurotus spp. in biotechnology. PeerJ. 2019 Mar 29;7: e6664. doi: 10.7717/peerj.6664.
- Wawoczny A, Przypis M, Gillner D. Enzymatic valorization of lignocellulosic biomass. Sustainability. 2023;15(24):16726. doi:10.3390/su152416726
- Nawafleh N, Al-Oqla FM. Applications of bio-composites in electronics. Polymers. 2024;9(12):3601-3620.
- Kumar V, Pallavi P, Sen SK, Raut S. Textile dye degradation by white rot fungi. Water Environ Res. 2024;96(1): e10959. doi:10.1002/wer.10959
- Aghaee M, et al. Laccase-MOF composites for pollutant remediation. Bioresour Technol. 2024. doi: 10.1016/j.biortech.2024.131072
- Das J, Badak M, Singh RK. White rot fungi-derived laccase innovations. In: Gupta J, Verma A, eds. Microbiology-2.0. Springer; 2024:285-306.
- Wang H, et al. Laccase immobilization for emerging pollutant degradation. J Environ Manage. 2024. doi: 10.1016/j.jenvman.2024.120984
- Akram F, Ashraf S, Haq IU, Shah FI, Aqeel A. Industrial applications of bacterial laccases. Appl Biochem Biotechnol. 2022;194(5):2336-2356. doi:10.1007/s12010-021-03781-9
- T E, et al. Free and immobilized biocatalysts in paper effluent treatment. Sep Purif Rev. 2024. doi:10.1080/15422119.2024.2382313
- Wijerathna P, Chen X, Qiu R, et al. Natural products from mangrove fungi. Molecules. 2026;31(2):261. doi:10.3390/molecules31020261
- Garkani Nejad F, Tajik S, Beitollahi H, Sheikhshoaie I. Magnetic nanomaterial-based biosensors. Talanta. 2021; 228:122075. doi: 10.1016/j.talanta.2020.122075
- Ogunsile A, Songnaka N, Sawatdee S, et al. Anti-MRSA peptides from Brevibacillus. PeerJ. 2023;11:e16143. doi:10.7717/peerj.16143
- Mate DM, Alcalde M. Laccase as a multi-purpose biocatalyst. Microb Biotechnol. 2017;10(6):1457-1467. doi:10.1111/1751-7915.12724
- Hossain, M.S., Alam, S., Mwamburi, L., Rahman, A., Binduraz, B., Roy, S., Sultana, M.T., Ghosh, S., & Haque, M.A. (2025). Decoding genome, phylogenetic insights, plant-beneficial genetic repertoire, and in silico pesticide biodegradation pathways of endophytic strain serratia sp hstu abk35. J. Biosci. Public Health, 2(1), 30-54. https://doi.org/10.5455/JBPH.2025.20
- Sharma K, Singh R, Yadav P, et al. Bacterial laccase-mediated dye biodegradation. Biologia. 2025;80(8):2145-2157.
- Beltrán-Flores E, et al. Effect of oxygen on white-rot fungi degradation. J Water Process Eng. 2023; 55:104105. doi: 10.1016/j.jwpe.2023.104105
- Singha S, Panda T. Optimization of laccase fermentation. Prep Biochem Biotechnol. 2015;45(4):307-335. doi:10.1080/10826068.2014.923453
- Enaime G, et al. Olive mill wastes: from waste to resources. Environ Sci Pollut Res. 2024;31(14):20853-20880.
- Rodríguez-Couto S. Fungal laccase applications. In: Yadav AN, et al., eds. White Biotechnology Through Fungi. Springer; 2019:429-457.
- Cheute VM, et al. Biotransformation by Pycnoporus spp. Biomass. 2024; 4:313-328. doi:10.3390/biomass4020015
- Baghel S, Sahariah BP, Anandkumar J. Bioremediation of pulp industry effluent. In: Shah M, Banerjee A, eds. Industrial Effluent Treatment. Springer; 2020:261-278.
- Khatua S. Myco-enzymes for xenobiotic degradation. In: Roy S, Mandal V, eds. Plant-Microbe Interaction. Springer; 2025:549-584.
- Borah M, et al. Microbial strategies for agricultural waste remediation. In: Shah MP, ed. Waste Water Treatment Research. Elsevier; 2025:297-321.
- Knežević A, Stajić M, et al. Bioactivity of Trametes species. PLoS One. 2018;13(8):e0203064. doi: 10.1371/journal.pone.0203064
- Vasile C, Baican M. Lignins as renewable biopolymers. Polymers. 2023;15. doi:10.3390/polym15153177
- Mayolo-Deloisa K, González-González M, Rito-Palomares M. Laccases in food industry. Front Bioeng Biotechnol. 2020;8. doi:10.3389/fbioe.2020.000XX (verify)
- Nunes CS, Kunamneni A. Laccases: properties and applications. In: Nunes CS, Kumar V, eds. Enzymes in Human and Animal Nutrition. Academic Press; 2018:133-161.
- Servili M, Selvaggini R, et al. Removal of phenols from grape must. Am J Enol Vitic. 2000;51(4):357-361.
- Minussi RC, Pastore GM, Durán N. Applications of laccase in food industry. Trends Food Sci Technol. 2002;13(6):205-216. doi:10.1016/S0924-2244(02)00125-3
- Sugano Y, Yoshida T. DyP-type peroxidases: advances and perspectives. Int J Mol Sci. 2021; 22:5556. doi:10.3390/ijms22115556
- Konstantinou IK, Albanis TA. TiO₂-assisted photocatalytic degradation of azo dyes in aqueous solution: kinetic and mechanistic investigations: a review. Appl Catal B Environ. 2004;49(1):1-14.
- Catucci G, et al. Biochemical features of dye-decolorizing peroxidases: current impact on lignin degradation. Biotechnol Appl Biochem. 2020;67(5):751-759.
- Sousa AC, Martins LO, Robalo MP. Laccase-catalysed homocoupling of primary aromatic amines towards the biosynthesis of dyes. Adv Synth Catal. 2013;355(14-15):2908-2917.
- Zerva A, et al. Applications of microbial laccases: patent review of the past decade (2009–2019). Catalysts. 2019; 9:1023. doi:10.3390/catal9121023
- Egbewale SO, Kumar A, Mokoena MP, et al. Purification, characterization and three-dimensional structure prediction of multicopper oxidase laccases from Trichoderma lixii FLU1 and Talaromyces pinophilus FLU12. Sci Rep. 2024; 14:13371. doi:10.1038/s41598-024-63959-z
- Iqbal M, Farooq S, Hussain A. Advances in protein- and lipid-based materials for cosmetic applications. In: Bandara N, Ullah A, eds. Functional Materials from Lipids and Proteins. Royal Society of Chemistry; 2024.
- Cardullo N, Muccilli V, Tringali C. Laccase-mediated synthesis of bioactive natural products and their analogues. ChemBioChem. 2022;23(12): e202100700. doi:10.1002/cbic.202100700
- Hahn V, Davids T, Lalk M, et al. Potential of the enzyme laccase for the synthesis and derivatization of antimicrobial compounds. World J Microbiol Biotechnol. 2023;39(4):98. doi:10.1007/s11274-023-03539-x
- Singh AK, Bilal M, Iqbal HMN, et al. Laccase-based biocatalytic systems application in sustainable degradation of pharmaceutically active contaminants. J Hazard Mater. 2025; 484:136384. doi: 10.1016/j.jhazmat.2024.136384
- Xu L, Wang Y, Zhang Z, et al. Degradation of tetracycline by laccase–mediator system using tea polyphenols as mediator. Catalysts. 2025;15(10):952. doi:10.3390/catal15100952
- Chmelová D, Ondrejovič M. Laccases as effective tools in the removal of pharmaceutical micropollutants from wastewater. Life (Basel). 2024;14(2):230. doi:10.3390/life14020230
- Ouyang B, Zhang X, Guo Y, et al. Efficient removal of sulfonamides and tetracyclines by a laccase from Lysinibacillus fusiformis. J Environ Chem Eng. 2022;10(6):108682. doi: 10.1016/j.jece.2022.108682
- Zdarta J, Jankowska K, Bachosz K, et al. Degradation of tetracyclines and sulfonamides by immobilized laccase and the effect on residual antibiotic activity. J Chem Technol Biotechnol. 2018;93(12):3415-3424. doi:10.1002/jctb.5697
- Hahn V, Sayer C, et al. Enhanced laccase-mediated transformation of diclofenac and other non-phenolic pollutants. Environ Sci Pollut Res. 2018;25(36):36088-36099.
- Roy MK, Roy S, Binduraz B, Afrin L, Haque MA. Pesticide-associated health and environmental risks and the role of biofertilizers in sustainable agriculture. J. Biosci. Public Health. 2025;1(3):16-27. doi:10.5455/JBPH.2025.12
- Pezzella C, Guarino L, Piscitelli A. How to enjoy laccases. Cell Mol Life Sci. 2015;72(5):923-940.
- Oudia A, Queiroz J, Simões R. The influence of operating parameters on the biodelignification of Eucalyptus globulus kraft pulps in a laccase–violuric acid system. Appl Biochem Biotechnol. 2008;149(1):23-32.
- Gutiérrez A, del Río JC, Rencoret J, Ibarra D, Martínez AT. Main lipophilic extractives in different paper pulp types can be removed using the laccase-mediator system. Appl Microbiol Biotechnol. 2006;72(4):845-851. doi:10.1007/s00253-006-0346-1
- Rodríguez-Couto S, Toca Herrera JL. Industrial and biotechnological applications of laccases: a review. Biotechnol Adv. 2006;24(5):500-513.
- Fillat Ú, et al. Laccases as a potential tool for the efficient conversion of lignocellulosic biomass: a review. Fermentation. 2017; 3:17. doi:10.3390/fermentation3020017
- Justino C, et al. Evaluation of tertiary treatment by fungi, enzymatic and photo-Fenton oxidation on the removal of phenols from a kraft pulp mill effluent: a comparative study. Biodegradation. 2011;22(2):267-274.
- Sobti R, Sharma A, Soni SK. Applications of biotechnological techniques in mitigating environmental concerns. In: Genomic, Proteomics, and Biotechnology. CRC Press; 2022:249-312.
- MacVittie K, Conlon T, Katz E. A wireless transmission system powered by an enzyme biofuel cell implanted in an orange. Bioelectrochemistry. 2015; 106:28-33.
- Chen T, et al. A miniature biofuel cell. J Am Chem Soc. 2001;123(35):8630-8631.
- Ellouze M, Sayadi S. White-rot fungi and their enzymes as a biotechnological tool for xenobiotic bioremediation. In: Management of Hazardous Wastes. 2016:103-120.
- Asad S, et al. Biotechnological potential of Ganoderma species: current progress and future prospects. N Z J Bot. 2025;63(5):2410-2469.
- El-Ramady H, et al. Green biotechnology of oyster mushroom (Pleurotus ostreatus L.): a sustainable strategy for myco-remediation and bio-fermentation. Sustainability. 2022; 14:3667. doi:10.3390/su14063667
- Zhang Y, et al. Application of eukaryotic and prokaryotic laccases in biosensor and biofuel cells: recent advances and electrochemical aspects. Appl Microbiol Biotechnol. 2018;102(24):10409-10423.
- Asaduzzaman M, et al. Degradation kinetics of lycopene from red amaranth and preparation of winter melon jelly using this lycopene and comparison with commercial jelly. Heliyon. 2024;10(10).
- Begum K, et al. Potential allelopathic candidates for land use and possible sustainable weed management in South Asian ecosystem. Sustainability. 2019; 11:2649. doi:10.3390/su11092649
- Gan J, et al. Upgrading recalcitrant lignocellulosic biomass hydrolysis by immobilized cellulolytic enzyme-based nanobiocatalytic systems: a review. Biomass Convers Biorefin. 2024;14(4):4485-4509.
- Aggarwal S, Ikram S. A comprehensive review on bio-mimicked multimolecular frameworks and supramolecules as scaffolds for enzyme immobilization. Biotechnol Bioeng. 2023;120(2):352-398.
- Rath S, et al. Transforming lignin into value-added products: perspectives on lignin chemistry, lignin-based biocomposites, and pathways for augmenting ligninolytic enzyme production. Adv Compos Hybrid Mater. 2024;7(1):27.
- Bera K, Bhattacharya D, Mukhopadhyay M. Leveraging bacterial laccases to facilitate the decomposition of xenobiotic compounds: a review. 3 Biotech. 2024;14(12):317.
- Raheja Y, et al. Advancement in lignocellulolytic enzyme production: tailored strategies to overcome challenges in biomass hydrolysis. Syst Microbiol Biomanuf. 2025;5(3):948-968.
- Jiao M, Chen W, Jiang X, Huang D, Jiang Y. Recent advances in laccase production: challenges and future perspectives. J Microbiol Biotechnol. 2025;35: e240521. doi:10.4014/jmb.2410.10021
- Ndochinwa O, Wang Y, et al. Current status and emerging frontiers in enzyme engineering: an industrial perspective. Heliyon. 2024;10(11): e32673. doi: 10.1016/j.heliyon. 2024.e32673
- Farhan M, Hasani IW, Khafaga DSR, Ragab WM, Ahmed Kazi RN, Aatif M, Muteeb G, Fahim YA. Enzymes as Catalysts in Industrial Biocatalysis: Advances in Engineering, Applications, and Sustainable Integration. Catalysts. 2025; 15(9):891. https://doi.org/10.3390/catal15090891
- Jahanshahi DA, Tiyoula FN, Kavousi K, Beh-Afarin SR, Ariaeenejad S. Sustainable solutions through metagenomic laccases: industrial and environmental perspectives. Environ Technol Innov. 2025; 40:104568. doi: 10.1016/j.eti.2025.104568
- Haq IU, Saleem A, Chaudhary R, et al. Role of microbial laccases in valorization of lignocellulosic biomass to bioethanol. Front Bioeng Biotechnol. 2024; 12:1441075. doi:10.3389/fbioe.2024.1441075
- Ma Y, et al. Advances in immobilized enzyme systems for enhanced microplastic biodegradation: a review. Int J Biol Macromol. 2025;328(Pt 2):147656. doi: 10.1016/j.ijbiomac.2025.147656
- Adamian YA, et al. Recent developments in the immobilization of laccase on carbonaceous supports for environmental applications—a critical review. Front Bioeng Biotechnol. 2021; 9:778239. doi:10.3389/fbioe.2021.778239
- Amponsah L, Labus K, et al. Advancing enzyme immobilization for wastewater remediation: laccase-MOF-hydrogel biocatalyst system for sustainable dye decolorization. Mater Today Sustain. 2025. doi: 10.1016/j.mtsust.2025.100492
- Chen X, Mosier NS, et al. Valorization of lignin from aqueous-based lignocellulosic biorefineries. Trends Biotechnol. 2024;42(11):1348-1362. doi: 10.1016/j.tibtech.2024.07.001
- Lin C-X, Hsu H-H, Chang Y-H, Chen S-H, Lin S-B, Lou S-N, Chen H-H. Expanding the Applicability of an Innovative Laccase TTI in Intelligent Packaging by Adding an Enzyme Inhibitor to Change Its Coloration Kinetics. Polymers. 2021; 13(21):3646. https://doi.org/10.3390/polym13213646
- Global Laccase Market Analysis and Forecast 2026-2032. Published Jan 02, 2026. Accessed; April 12, 2026. https://www.marketresearch.com/APO-Research-Inc-v4273/Global-Laccase-Forecast-43749055
- Islam MS. The Emerging Role of Microcredentials in Higher Education: Advancing Public Health Learning and Beyond. J Biosci Public Health. 2025;1(1):43-54. doi: 10.5455/JBPH.2025.04
 access
access