- Kabir KH, Aftab S. Exploring management strategies for freshwater wetlands: policy options for southwest coastal region in Bangladesh. Asian Dev Policy Rev. 2017;5(2):70-80.
- Siddiquee SA, Hoque ME. Wetland conservation in context of climate induced changes: Bangladesh perspective. J Econ Sustain Dev. 2011;2(3):1-8.
- Sohel MSI, Mukul SA, Chicharo L. A new ecohydrological approach for ecosystem service provision and sustainable management of aquatic ecosystems in Bangladesh. Ecohydrol Hydrobiol. 2015;15(1):1-12. doi: 10.1016/j.ecohyd.2014.12.001
- Islam MM. Unravelling the complexities of wetland agriculture, climate change, and coping mechanisms: an integrative review using economics and satellite approaches. Environ Dev Sustain. 2024:1-33. doi:10.1007/s10668-024-04579-0
- Boral Doal D, Mannan MA, Rahman MM, Higuchi H. Groundwater use efficiency for sustainable agriculture: prospects in Bangladesh. Big Data Water Resour Eng. 2023.
- Khan ZH, Islam MS, Akhter S, Hasib MR, Sutradhar A, Timsina J, Krupnik TJ, Schulthess U. Can crop production intensification through irrigation be sustainable? An ex-ante impact study of the south-central coastal zone of Bangladesh. PLOS Water. 2024;3(2): e0000153. doi: 10.1371/journal.pwat.0000153
- Shamsudduha M, Taylor RG, Haq MI, Nowreen S, Zahid A, Ahmed KMU. The Bengal water machine: quantified freshwater capture in Bangladesh. Science. 2022;377(6612):1315-1319. doi:10.1126/science. abn2508
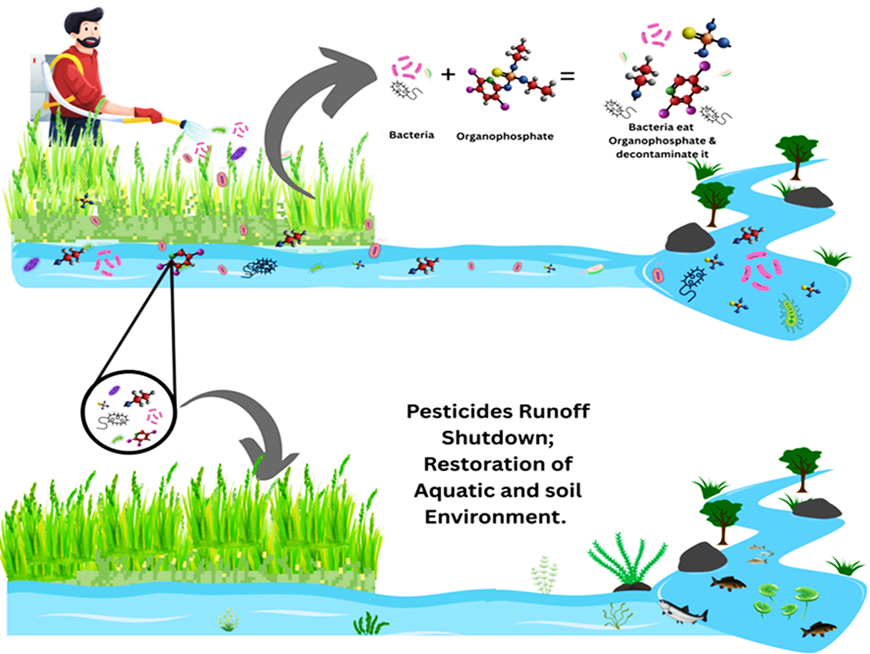
- Hasanuzzaman M, Rahman MA, Islam MS, Salam MA, Nabi MR. Pesticide residues analysis in water samples of Nagarpur and Saturia Upazila, Bangladesh. Appl Water Sci. 2018;8(1):8. doi:10.1007/s13201-018-0664-1
- Laurent A, Faragò M, Secchi M. Globally-differentiated land use flow inventories for life cycle impact assessment. In: SETAC Europe 27th Annual Meeting: Environmental Quality Through Transdisciplinary Collaboration; 2017:167.
- Khatib I, Rychter P, Falfushynska H. Pesticide pollution: detrimental outcomes and possible mechanisms of fish exposure to common organophosphates and triazines. J Xenobiot. 2022;12(3):236-265. doi:10.3390/jox12030018
- Pesce S, Mamy L, Sanchez W, et al. Main conclusions and perspectives from the collective scientific assessment of the effects of plant protection products on biodiversity and ecosystem services along the land–sea continuum in France and French overseas territories. Environ Sci Pollut Res. 2025;32(6):2757-2772. doi:10.1007/s11356-024-33026-1
- Villamarín C, Cañedo-Argüelles M, Carvajal-Rebolledo C, Ríos-Touma B. Effects of pesticides on the survival of shredder Nectopsyche sp. (Trichoptera) and leaf decomposition rates in tropical Andes: a microcosm approach. Toxics. 2022;10(12):720. doi:10.3390/toxics10120720
- Stehle S, Schulz R. Agricultural insecticides threaten surface waters at the global scale. Proc Natl Acad Sci U S A. 2015;112(18):5750-5755. doi:10.1073/pnas.1500232112
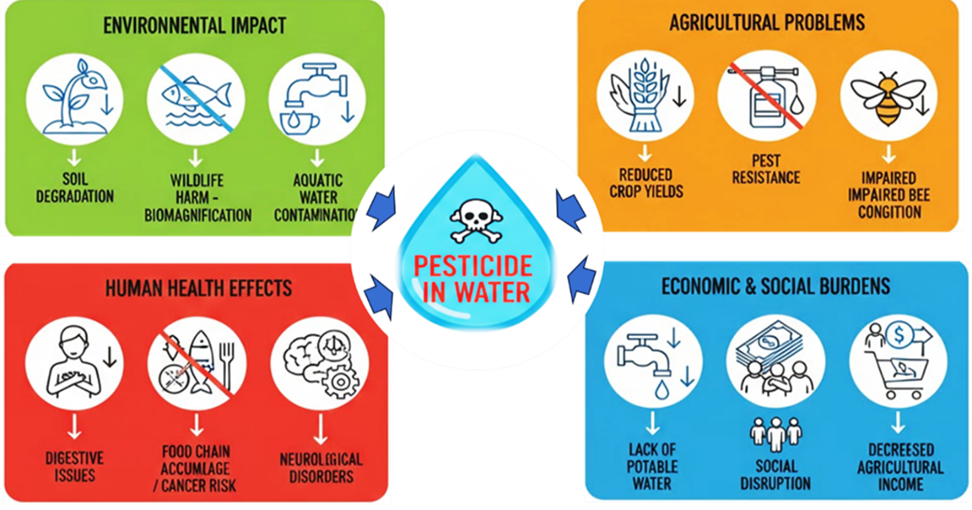
- Zhou W, Li M, Achal V. A comprehensive review on environmental and human health impacts of chemical pesticide usage. Emerg Contam. 2025;11(1):100410. doi: 10.1016/j.emcon.2024.100410
- Kadiru S, Patil S, D’Souza R. Effect of pesticide toxicity in aquatic environments: a recent review. Int J Fish Aquat Stud. 2022;10(3):113-118.
- Brasseur MV, Buchner D, Mack L, Schreiner VC, Schäfer RB, Leese F, Mayer C. Multiple stressor effects of insecticide exposure and increased fine sediment deposition on gene expression profiles of two freshwater invertebrate species. Environ Sci Eur. 2023;35(1):81. doi:10.1186/s12302-023-00785-6
- Góngora CE, Silva MDC. Sustainable strategies for the control of crop diseases and pests to reduce pesticides. Agronomy. 2024;14(9):2158. doi:10.3390/agronomy14092158
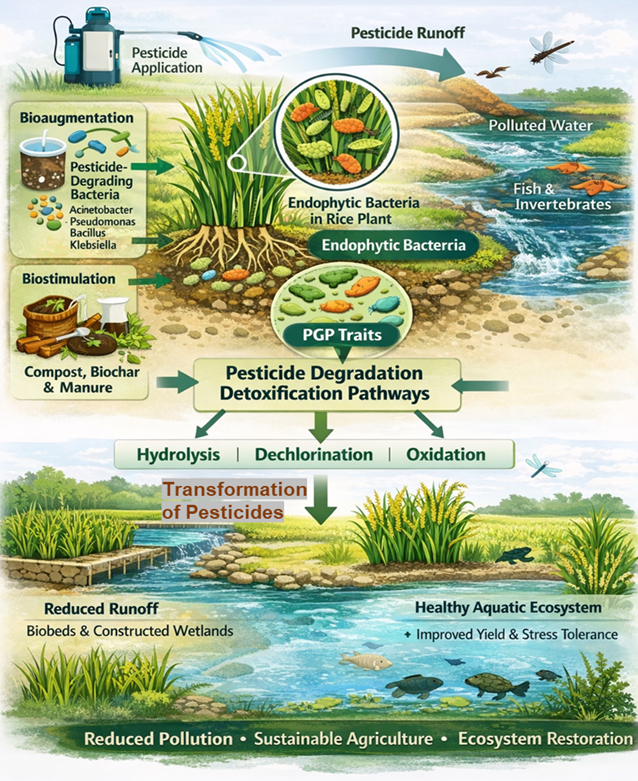
- Datta S, Singh S, Kumar V, Dhanjal DS, Sidhu GK, Amin DS, Singh J. Endophytic bacteria in xenobiotic degradation. In: Verma SK, White JF, eds. Microbial Endophytes. Woodhead Publishing; 2020:125-156. doi:10.1016/B978-0-12-818734-0.00006-6
- Manganyi MC, Dikobe TB, Maseme MR. Exploring the potential of endophytic microorganisms and nanoparticles for enhanced water remediation. Molecules. 2024;29(12):2858. doi:10.3390/molecules29122858
- Feng NX, Yu J, Zhao HM, Cheng YT, Mo CH, Cai QY, Wong MH. Efficient phytoremediation of organic contaminants in soils using plant–endophyte partnerships. Sci Total Environ. 2017; 583:352-368. doi: 10.1016/j.scitotenv.2017.01.075
- Sustainable crop production. In: Woodhead Publishing Series in Food Science, Technology and Nutrition. Elsevier; 2023:237-271. doi:10.1016/B978-0-323-85702-4.00019-4
- Chen WM, Tang YQ, Mori K, Wu XL. Distribution of culturable endophytic bacteria in aquatic plants and their potential for bioremediation in polluted waters. Aquat Biol. 2012;15(2):99-110. doi:10.3354/ab00418
- Obi LU, Akinyemi MO, Alayande KA, Adeleke RA. Application of endophytes in bioremediation, biotransformation, and water disinfection for irrigation systems. In: Enhancing Water and Food Security Through Improved Agricultural Water Productivity. Springer Nature Singapore; 2025:117-137.
- Salam MTB, Ito K, Kataoka R. Biodegradation of nitenpyram by endophytic bacterium Bacillus thuringiensis strain NIT-2 isolated from neonicotinoid-treated plant samples. J Pestic Sci. 2024;49(2):94-103. doi:10.1584/jpestics. D23-046
- Faridy N, Torabi E, Pourbabaee AA, Osdaghi E, Talebi K. Efficacy of novel bacterial consortia in degrading fipronil and thiobencarb in paddy soil. Front Microbiol. 2024; 15:1366951. doi:10.3389/fmicb.2024.1366951
- Prodhan MY, Rahman MB, Rahman A, Akbor MA, Ghosh S, Nahar MNEN, et al. Characterization of growth-promoting activities of consortia of chlorpyrifos-mineralizing endophytic bacteria naturally harboring in rice plants a potential biostimulant to develop safe and sustainable agriculture. Microorganisms. 2023;11(7):1821. doi:10.3390/microorganisms11071821
- Rahaman MM, Islam KS, Jahan M. Rice farmers’ knowledge of the risks of pesticide uses in Bangladesh. J Health Pollut. 2018;8(20):181203. doi:10.5696/2156-9614-8.20.181203
- Sumon KA, Rico A, Ter Horst MM, Van den Brink PJ, Haque MM, Rashid H. Risk assessment of pesticides used in rice-prawn concurrent systems in Bangladesh. Sci Total Environ. 2016; 568:498-506. doi: https://doi.org/10.1016/j.scitotenv.2016.06.014
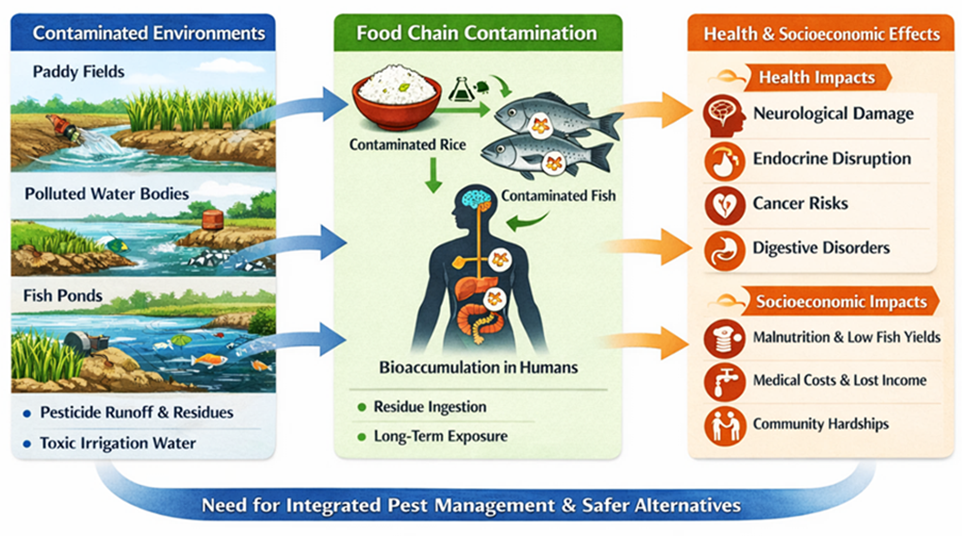
- Mithu MMU, Shormela SA, Abdullah ATM, et al. Exploring heavy metal bioaccumulation in vegetables: unraveling environmental pollutants' impact on agricultural produce and human health. Biol Trace Elem Res. 2025. doi:10.1007/s12011-025-04871-z
- Braschi I, Blasioli S, Lavrnić S, Buscaroli E, Di Prodi K, Solimando D, Toscano A. Removal and fate of pesticides in a farm constructed wetland for agricultural drainage water treatment under Mediterranean conditions (Italy). Environ Sci Pollut Res. 2022;29(5):7283-7299. doi:10.1007/s11356-021-16033-4
- da Silva Santarossa MA, Coleone AC, de Mello NP, Ignácio NF, Machado AA, Marques Silva JR, Velini ED, Machado Neto JG. Contamination of fee-fishing ponds with agrochemicals used in sugarcane crops. SN Appl Sci. 2020;2(9):1498. doi:10.1007/s42452-020-03274-0
- Mithu MMU, Shormela SA, Islam MS, Mubarak M. FTIR analysis of pesticide active ingredients into seasonal vegetables: ensuring food safety and raising awareness. J Glob Innov Agric Sci. 2025; 13:139-147. doi:10.22194/JGIAS/25.1463
- Ahmed MS, Prodhan MDH, Begum A, Afroze M, Dutta NK. Pesticide residue contamination in eggplant and hyacinth bean at eight different regions of Bangladesh. Asian-Australas J Food Saf Secur. 2024;8(2):67-74. doi:10.3329/aajfss. v8i2.77605
- Islam MA. Health risk assessment of pesticide residues in vegetables collected from northern part of Bangladesh. Food Res. 2020;4(6):2281-2288.
- Ahmed MS, Rahman MA, Begum A, Chowdhury AZ, Reza MS. Multi insecticide residue analysis in vegetables collected from different regions of Bangladesh. Asian-Australas J Biosci Biotechnol. 2016;1(3):547-551. doi:10.3329/aajbb. v1i3.64036
- Ahmed MS, Begum A, Prodhan MDH, Afroze M, Sarker D. Organophosphorus pesticide residues detected in eggplant and tomato samples collected from different regions of Bangladesh. Asian-Australas J Food Saf Secur. 2021;5(1):27-31. doi:10.3329/aajfss. v5i1.55015
- Miah MH, Chand DS, Malhi GS. Selected river pollution in Bangladesh based on industrial growth and economic perspective: a review. Environ Monit Assess. 2023;195(1):98. doi:10.1007/s10661-022-10663-y
- Parvin F, Haque MM, Tareq SM. Recent status of water quality in Bangladesh: a systematic review, meta-analysis and health risk assessment. Environ Chall. 2022; 6:100416. doi: 10.1016/j.envc.2021.100416
- Wakiluzzaman SM. (2025). Public Health Implications of Pesticide Use in Bangladesh: A Comprehensive Review. International Journal of Research and Scientific Innovation, 259. https://doi.org/10.51244/ijrsi.2025.121500025p
- Acharya S, Upadhayay HR, Houbraken M, Bajracharya RM, Spanoghe P. Occurrence of unapproved pesticides and their ecotoxicological significance for an agriculturally influenced reservoir and its tributaries in Nepal. Water Air Soil Pollut. 2023;234(9):565. doi:10.1007/s11270-023-06570-8
- Hasan MM, Farouque MG, Sarker MA. An assessment of using eco-friendly crop production practices by the project beneficiaries and non-beneficiaries in Bangladesh. Discover Agric. 2024;2(1):21. doi:10.1007/s44279-024-00034-1
- Salma U, Shafiujjaman M, Al Zahid M, Faruque MH, Habibullah-Al-Mamun M, Hossain A. Widespread use of antibiotics, pesticides, and other aqua-chemicals in finfish aquaculture in Rajshahi District of Bangladesh. Sustainability. 2022;14(24):17038. doi:10.3390/su142417038
- Ndayambaje B, Amuguni H, Coffin-Schmitt J, Sibo N, Ntawubizi M, VanWormer E. Pesticide Application Practices and Knowledge among Small-Scale Local Rice Growers and Communities in Rwanda: A Cross-Sectional Study. Int J Environ Res Public Health. 2019 Nov 28;16(23):4770. doi: 10.3390/ijerph16234770.
- Yang X, Silva V, Tang DW. Pesticide transport under runoff-erosion potentially dominated by small sediments: a glyphosate and AMPA experiment. J Hydrol. 2025;133633. doi: 10.1016/j.jhydrol.2025.133633
- Nurullah ABM, Khatun MS, Ritchie L. Industrial wastewater disposal and its socio-environmental consequences: evidence from the Uttara Export Processing Zone, Bangladesh. Sustainability. 2025;17(17):7716. doi:10.3390/su17177716
- Ari IRD, Wijatmiko I, Santosa H, Prayitno G. Empowering smart health cities through innovative water management strategies. Int J Sustain Dev Plan. 2024;19(8). doi:10.18280/ijsdp.190816
- Yadav SC. Water pollution: the problems and solutions. Science Insights. 2024;44(2):1245-1251. doi:10.15354/si.24.re905
- De Carvalho FG, Loyau A, Kelly-Irving M, Schmeller DS. Aquatic ecosystem indices, linking ecosystem health to human health risks. Biodivers Conserv. 2025;34(3):723-767. doi:10.1007/s10531-025-03010-3
- Gupta LK, Pandey M, Raj PA, Shukla AK. Fine sediment intrusion and its consequences for river ecosystems: a review. J Hazard Toxic Radioact Waste. 2023;27(1):04022036. doi:10.1061/(ASCE)HZ.2153-5515.0000729
- Khan WA, Ali S, Shah SA. Water pollution: sources and its impact on human health, control and managing. J Int Coop Dev. 2022;5(1):69. doi:10.36941/jicd-2022-0005
- Yusof NADM, Voon NSMF, Nordin N, Karuppiah K, Jamil PASM. Pollution in modern cities: a comprehensive overview with focus on eye health and medical imaging. In: Environmental Sciences: Urban Pollution‑Environmental Challenges in Healthy Modern Cities. IntechOpen. 2024. doi:10.5772/intechopen.1007548
- Sahoo S, Baral N, Nayak A, Naik A, Behera D, Mahapatra M, Sahoo JP. Effect of pesticides on human health and biodiversity: a comprehensive insight. Bhartiya Krishi Anusandhan Patrika. 2024;39(2):115-124. doi:10.18805/BKAP734
- Wang J, Wang Z, Dou Y, Cong J, Sun H, Wang L, Duan Z. Ecological risk assessment for typical organophosphorus pesticides in surface water of China based on a species sensitivity distribution model. Sci Total Environ. 2024; 913:169805. doi: 10.1016/j.scitotenv.2023.169805
- Nehul JN. Environmental impact of pesticides: toxicity, bioaccumulation and alternatives. Environ Rep. 2025;7(2):14‑21. doi:10.51470/ER.2025.7.2.14
- Marques MBL, Brunetti IA, Faleiros CA, da Cruz C, Iqbal HM, Bilal M, Américo-Pinheiro JHP. Ecotoxicological assessment and environmental risk of the insecticide chlorpyrifos for aquatic neotropical indicators. Water Air Soil Pollut. 2021;232(10):428. doi:10.1007/s11270-021-05369-9
- Barb, J. S. (2025). Evaluation of Chlorpyriphos as an Organophosphate Pesticide on Hematology, Blood Biochemistry, Antioxidant Capacity, Oxidative Stress, and Histopathological Changes in.
- Mathiyazhagan N, Kesavan D, Monu V, Manickam S, Hamed AG, Sabariswaran K. Assessing the ecological impact of pesticides/herbicides on algal communities: a comprehensive review. Aquat Toxicol. 2024; 268:106851. doi: 10.1016/j.aquatox.2024.106851
- Baudrot V, Fritsch C, Perasso A, Banerjee M, Raoul F. Effects of contaminants and trophic cascade regulation on food chain stability: Application to cadmium soil pollution on small mammals–raptor systems. Ecol Model. 2018; 382:33-42. doi: 10.1016/j.ecolmodel.2018.05.002
- Carrick HJ, Rohr JR, Hoverman JT, Mahon MB, Hudson PJ, Rumschlag SL, Raffel TR. Consistent effects of pesticides on community structure and ecosystem function in freshwater systems. 2020. doi:10.1038/s41467-020-20192-2
- Okogwu OI, Elebe FA, Nwonumara GN. Combinations of cypermethrin and dimethoate alter behavior, hematology and histology of African Catfish, Clarias gariepinus. Environ Anal Health Toxicol. 2022;37(4): e2022028. doi:10.5620/eaht.2022028
- Kumar P, Kumar R, Thakur K, Mahajan D, Brar B, Sharma D, Sharma AK. Impact of pesticides application on aquatic ecosystem and biodiversity: a review. Biol Bull. 2023;50(6):1362-1375. doi:10.1134/S1062359023601386
- Ramasre JR, Kashyap N, Chandravanshi S, Mishra S, Baidya S, Lal J, Dhruve D. Endocrine disrupting chemicals and their harmful effects in fish: a comprehensive review. Int J Adv Biochem Res. 2024;8(3S):5-11. doi:10.33545/26174693. 2024.v8. i3Sa.677
- Yang L, Li L, Kong Z, Zhang Y, Francis F, Fan B, Li M. Environmental reproductive toxicity of common pesticides: mechanisms and implications throughout the zebrafish life cycle. Ecotoxicol Environ Saf. 2025; 304:119029. doi: 10.1016/j.ecoenv.2025.119029
- de Arcaute CR, Larramendy ML, Soloneski S. Genotoxicity by long-term exposure to the auxinic herbicides 2,4-dichlorophenoxyacetic acid and dicamba on Cnesterodon decemmaculatus (Pisces: Poeciliidae). Environ Pollut. 2018; 243:670-678. doi: 10.1016/j.envpol.2018.09.040
- Hadeed MN, Castiglione CL, Saleem S, Chammout DH, Muskovac MD, Crile KG, Belanger RM. Environmentally relevant atrazine exposure leads to increases in DNA damage and changes in morphology in the hepatopancreas of crayfish (Faxonius virilis). Environ Adv. 2022; 10:100320. doi: 10.1016/j.envadv.2022.100320
- Li Z, Köhler H-R, Triebskorn R. Environmental Drivers of Pesticide Toxicity: Temperature and pH Shift Azoxystrobin’s Effects on Zebrafish (Danio rerio) Early Development. Environments. 2025; 12(9):334. https://doi.org/10.3390/environments12090334
- Abuqamar SF, El-Saadony MT, Alkafaas SS, Elsalahaty MI, Elkafas S, Mathew BT, El-Tarabily KA. Ecological impacts and management strategies of pesticide pollution on aquatic life and human beings. Mar Pollut Bull. 2024; 206:116613. doi: 10.1016/j.marpolbul.2024.116613
- Fritsch C, Berny P, Crouzet O, Le Perchec S, Coeurdassier M. Wildlife ecotoxicology of plant protection products: knowns and unknowns about the impacts of currently used pesticides on terrestrial vertebrate biodiversity. Environ Sci Pollut Res. 2025;32(6):2893-2955. doi:10.1007/s11356-024-33026-1
- Wan NF, Fu L, Dainese M, Kiær LP, Hu YQ, Xin F, Goulson D, Woodcock B, Vanbergen A, Spurgeon D, Scherber C. The impact of pesticides on non-target organisms. 2023. doi:10.21203/rs.3.rs-2549816/v1
- Hossain MM, Jahan I, Dar MA, Dhanavade MJ, Mamtaz AFB, Maxwell SJ, Zhu D. A review of potentially toxic elements in sediment, water, and aquatic species from the river ecosystems. Toxics. 2024;13(1):26. doi:10.3390/toxics13010026
- Sumon KA, Rashid H, Peeters ETHM, Bosma RH, Van den Brink PJ. Environmental monitoring and risk assessment of organophosphate pesticides in aquatic ecosystems of north-west Bangladesh. Chemosphere. 2018; 206:92-100. doi: 10.1016/j.chemosphere.2018.04.167
- Tóth G, Háhn J, Szoboszlay S, Harkai P, Farkas M, Radó J, Göbölös B, Kaszab E, Szabó I, Urbányi B, Kriszt B. Spatiotemporal analysis of multi-pesticide residues in the largest Central European shallow lake, Lake Balaton, and its sub-catchment area. Environ Sci Eur. 2022;34(1):50. doi:10.1186/s12302-022-00630-2
- Acharya LK, Paramaguru PK, Tripathi K, Bhoi TK, Seth P, Birah A. Pesticide contamination in groundwater: processes, risks, and mitigation strategies. Discov Agric. 2025;3(1):152. doi:10.1007/s44279-025-00337-x
- Dehghani MH, Ahmadi S, Ghosh S, Khan MS, Othmani A, Khanday WA, Ansari K. Sustainable remediation technologies for removal of pesticides as organic micro-pollutants from water environments: a review. Appl Surf Sci Adv. 2024; 19:100558. doi: 10.1016/j.apsadv.2023.100558
- Schmidt SN, Burgess RM. Evaluating partition-based sampling for predicting the bioaccumulation of hydrophobic organic contaminants by higher trophic level organisms. In: Proceedings of the Society of Environmental Toxicology and Chemistry (SETAC) North America 39th Annual Meeting; 2018; Sacramento, CA. SETAC; 2018:259-260.
- Doody DG, Withers PJ, Dils RM, McDowell RW, Smith V, McElarney YR, Dunbar M, Daly D. Optimizing land use for the delivery of catchment ecosystem services. Front Ecol Environ. 2016;14(6):325-332. doi:10.1002/fee.1296
- Morin S, Artigas J. Twenty years of research in ecosystem functions in aquatic microbial ecotoxicology. Environ Toxicol Chem. 2023;42(9):1867-1888. doi:10.1002/etc.5708
- Salis RK, Schreiner VC, Rozenberg A, Ohler K, Baudy-Groh P, Schäfer RB, Leese F. Effects of fungicides on aquatic fungi and bacteria: a comparison of morphological and molecular approaches from a microcosm experiment. Environ Sci Eur. 2023;35(1):62. doi:10.1186/s12302-023-00768-7
- Staley ZR, Harwood VJ, Rohr JR. A synthesis of the effects of pesticides on microbial persistence in aquatic ecosystems. Crit Rev Toxicol. 2015;45(10):813-836. doi:10.3109/10408444.2015.1065471
- Barbieri MV, Peris A, Postigo C, Moya-Garces A, Monllor-Alcaraz LS, Rambla-Alegre M, de Alda ML. Evaluation of the occurrence and fate of pesticides in a typical Mediterranean delta ecosystem (Ebro River Delta) and risk assessment for aquatic organisms. Environ Pollut. 2021; 274:115813. doi: 10.1016/j.envpol.2020.115813
- McKnight US, Rasmussen JJ, Kronvang B, Binning PJ, Bjerg PL. Sources, occurrence and predicted aquatic impact of legacy and contemporary pesticides in streams. Environ Pollut. 2015; 200:64-76. doi: 10.1016/j.envpol.2015.02.015
- Shrivastav G, Dubey A, Kumari M, Shukla T. Advances in pharmacognosy for modern drug discovery and development. J Biosci Public Health. 2025;1(3):45-63. https://doi.org/10.5455/JBPH.2025.14
- Bighiu MA, Goedkoop W. Interactions with freshwater biofilms cause rapid removal of common herbicides through degradation–evidence from microcosm studies. Environ Sci Process Impacts. 2021;23(1):66-72. doi: 10.1039/d0em00394h
- Borreca A, Vuilleumier S, Imfeld G. Combined effects of micropollutants and their degradation on prokaryotic communities at the sediment–water interface. Sci Rep. 2024;14(1):16840. doi:10.1038/s41598-024-67308-y
- Elzakey EM, El-Sabbagh SM, Eldeen EESN, Adss IAA, Nassar AMK. Bioremediation of chlorpyrifos residues using some indigenous species of bacteria and fungi in wastewater. Environ Monit Assess. 2023;195(6):779. doi:10.1007/s10661-023-11341-3
- Michel C, Baran N, André L, Charron M, Joulian C. Side effects of pesticides and metabolites in groundwater: impact on denitrification. Front Microbiol. 2021; 12:662727. doi:10.3389/fmicb.2021.662727
- Stenstrom JR, Kreuger J, Goedkoop W. Pesticide mixture toxicity to algae in agricultural streams: field observations and laboratory studies with in situ samples and reconstituted water. Ecotoxicol Environ Saf. 2021; 215:112139. doi: 10.1016/j.ecoenv.2021.112153
- Onyango J, van Bruggen JJA, Kitaka N, Simaika J, Irvine K. Effects of combined nutrient and pesticide exposure on algal biomass and Daphnia magna abundance. Environ Syst Res. 2024;13(1):1. doi:10.1186/s40068-023-00326-3
- Wijewardene L, Wu N, Qu Y, et al. Influences of pesticides, nutrients, and local environmental variables on phytoplankton communities in lentic small water bodies in a German lowland agricultural area. Sci Total Environ. 2021; 780:146481. doi: 10.1016/j.scitotenv.2021.146481
- Prabhu L, Pingali PL, Gerpacio RV. For cereal crops in Asia. In: Singh RP, ed. Agriculture and the Environment: Perspectives on Sustainable Rural Development. 1998:254.
- Bowers J. Sustainability, agriculture, and agricultural policy. Environ Plan A. 1995;27(8):1231-1243. doi:10.1068/a271231
- Yu Y, Li Y, Shen Z, Yang Z, Mo L, Kong Y, Lou I. Occurrence and possible sources of organochlorine pesticides (OCPs) and polychlorinated biphenyls (PCBs) along the Chao River, China. Chemosphere. 2014; 114:136-143. doi: 10.1016/j.chemosphere.2014.03.095
- Lamers M, Anyusheva M, La N, Nguyen VV, Streck T. Pesticide pollution in surface- and groundwater by paddy rice cultivation: A case study from Northern Vietnam. Clean Soil Air Water. 2011;39(4):356-361. doi: 10.1002/clen.201000268
- Duangchinda A, Anurugsa B, Hungspreug N. The use of organophosphate and carbamate pesticides on paddy fields and cholinesterase levels of farmers in Sam Chuk District, Suphan Buri Province, Thailand. Science & Technology Asia. 2014; 19:39-51.
- Chowdhury FR, Dewan G, Verma VR, Knipe DW, Isha IT, Faiz MA, Gunnell DJ, Eddleston M. Bans of WHO Class I Pesticides in Bangladesh-suicide prevention without hampering agricultural output. Int J Epidemiol. 2018 Feb 1;47(1):175-184. doi: 10.1093/ije/dyx157.
- Chau NDG, Sebesvari Z, Amelung W, Renaud FG. Pesticide pollution of multiple drinking water sources in the Mekong Delta, Vietnam: evidence from two provinces. Environ Sci Pollut Res DOI 10.1007/s11356-014-4034-x.
- Nakano Y, Miyazaki A, Yoshida T, Ono K, Inoue T. A study on pesticide runoff from paddy fields to a river in rural region-1: field survey of pesticide runoff in the Kozakura River, Japan. Water Research. 38 (13), 2004. 3017-3022. https://doi.org/10.1016/j.watres.2004.02.013.
- Naylor R, ed. 1996. Herbicides in Asian rice: transitions in weed management. Palo Alto (California): Institute for International Studies, Stanford University and Manila (Philippines): International Rice Research Institute. 270 p.
- Sudo M, Okubo T, Kaneki R. Paddy herbicide inputs in the entire river inflow reaching Lake Biwa, Japan. Limnology 6, 91–99 (2005). https://doi.org/10.1007/s10201-005-0145-x.
- Bunch MJ, Suresh VM, Kumaran TV. Environment and health aspects of pesticides use in Indian agriculture. Proceedings of the International Conference on Environment and Health. December 2003; 15:17.
- Nie J, Sun Y, Zhou Y, Kumar M, Usman M, Li J, Sun Y, Tsang DC. Bioremediation of water containing pesticides by microalgae: mechanisms, methods, and prospects for future research. Sci Total Environ. 2020; 707:136080. doi: 10.1016/j.scitotenv.2019.136080
- Kumar A, Goutam E, Thakur R, Geetika, Meshram S. Microbial degradation of pesticides in the environment. Microbial Applications for Environmental Sustainability. Singapore: Springer Nature Singapore; 2024:99-116.
- Mussali-Galante P, Castrejón-Godínez ML, Díaz-Soto JA, Vargas-Orozco ÁP, Quiroz-Medina HM, Tovar-Sánchez E, Rodríguez A. Biobeds, a microbial-based remediation system for the effective treatment of pesticide residues in agriculture. Agriculture. 2023;13(7):1289. doi:10.3390/agriculture13071289
- Aldas-Vargas A, van der Vooren T, Rijnaarts HH, Sutton NB. Biostimulation is a valuable tool to assess pesticide biodegradation capacity of groundwater microorganisms. Chemosphere. 2021; 280:130793. doi: 10.1016/j.chemosphere.2021.130793
- Onwona-Kwakye M, Plants-Paris K, Keita K, Lee J, van den Brink PJ, Hogarh JN, Darkoh C. Pesticides decrease bacterial diversity and abundance of irrigated rice fields. Microorganisms. 2020;8(3):318. doi:10.3390/microorganisms8030318
- Satish G, Ashokrao DM, Arun SK. Microbial degradation of pesticide: a review. African Journal of Microbiology Research. 2017;11(24):992-1012. doi:10.5897/AJMR2016.8402
- Abou Elatta A, El-Zawawy HAH, Afify AH, Hauka FIA. Degradation of chlorpyrifos by the cyanobacteria strains in rice fields. J Agric Chem Biotechnol. 2023;14(5):43-49. doi:10.21608/jacb.2023.202369.1050
- Aswathi A, Pandey A, Sukumaran RK. Rapid degradation of the organophosphate pesticide chlorpyrifos by a novel strain of Pseudomonas nitroreducens AR-3. Bioresour Technol. 2019; 292:122025. doi:10.1016/j.biortech.2019.122025
- Singh BK, Walker A. Microbial degradation of organophosphorus compounds. FEMS Microbiol Rev. 2006;30(3):428-471. doi:10.1111/j.1574-6976.2006.00018. x.
- Shoman R, Aggag S, Yacout M. Biodegradation of chlorpyrifos by some bacterial strains and screening their degraded genes. J Agric Chem Biotechnol. 2022;13(1):1-7. doi:10.21608/jacb.2022.116569.1016.
- Lakshmi CV, Kumar M, Khanna S. Biodegradation of chlorpyrifos, endosulfan and diazinon by Bacillus subtilis. World J Microbiol Biotechnol. 2009;25(7):1209-1216. doi:10.1007/s11274-009-0003-0.
- Bashan Y, de‑Bashan LE. How the plant growth‑promoting bacterium Azospirillum promotes plant growth — a critical assessment. Adv Agron. 2010; 108:77‑136.
- Mori T, Ohno H, Ichinose H, Kawagishi H, Hirai H. White-rot fungus Phanerochaete chrysosporium metabolizes chloropyridinyl-type neonicotinoid insecticides by an N-dealkylation reaction catalyzed by two cytochrome P450s. J Hazard Mater. 2021; 402:123831. doi: 10.1016/j.jhazmat.2020.123831
- Gong T, Liu R, Che Y, Xu X, Liu Y. Biodegradation of chlorpyrifos by Pseudomonas putida and characterization of degradation pathways. Chemosphere. 2018; 206:293-300. doi: 10.1016/j.chemosphere.2018.05.021.
- Xu WJ, Wan Q, Wang WF, Wang Y, Feng FY, Cheng JJ, Yu XY. Biodegradation of dibutyl phthalate by a novel endophytic Bacillus subtilis strain HB-T2 under in-vitro and in-vivo conditions. Environ Technol. 2022;43(13):1917-1926. doi:10.1080/09593330.2020.1858181
- Osadebe A, Patrick PU. Environmental detoxification potential of axenic and mixed cultures of Bacillus species on pesticides using an in vitro biodegradation assay. J. microbiol., biotechnol. food sci. 2023;12(5): e9272–e9272. doi:10.55251/jmbfs.9272
- Rahman MM, Khanom A, Biswas SK. Effect of pesticides and chemical fertilizers on the nitrogen cycle and functional microbial communities in paddy soils: Bangladesh perspective. Bull Environ Contam Toxicol. 2021;106(2):243-249. doi:10.1007/s00128-020-03092-5
- Bhagawati S, Bhattacharyya B, Bhattacharjee S, Devi EB, Manpoong NS, Das PP. Microbial bioremediation of pesticide residues: A review. Int J Curr Microbiol Appl Sci. 2020;9(4):1551-1561. doi:10.20546/ijcmas.2020.904.182
- Spina F, Cecchi G, Landinez-Torres A, Pecoraro L, Russo F, Wu B, Persiani AM. Fungi as a toolbox for sustainable bioremediation of pesticides in soil and water. Plant Biosyst. 2018;152(3):474-488. doi:10.1080/11263504.2018.1445130
- Haque MA, Simo, Prodhan MY, Ghosh S, Hossain MS, Rahman A, Haque MA. Enhanced rice plant (BRRI-28) growth at lower doses of urea caused by diazinon mineralizing endophytic bacterial consortia and explorations of relevant regulatory genes in a Klebsiella sp. strain HSTU-F2D4R. Arch Microbiol. 2023;205(6):231.
- Roy S, Sultana MT, Rahman A, Hossain MS, Binduraz B, Hanna T, Abir MAI, Rahman M, Abdullah-Al-Mamun M, Roy MK, Cho KM, Haque MA. Genomic and in silico characterization of Acinetobacter sp. strain HSTU-Asm16 with plant growth-promoting traits and diazinon biodegradation potential. J Biosci Public Health. 2026;2(1):68-95. doi:10.5455/JBPH.2026.02
- Das SR, Haque MA, Akbor MA, Abdullah-Al-Mamun M, Debnath GC, Hossain MS, Cho KM. Organophosphorus insecticides mineralizing endophytic and rhizospheric soil bacterial consortium influence eggplant growth-promotion. Arch Microbiol. 2022;204(3):199. doi:10.1007/s00203-022-02809-w
- Haque MA, Hossain MS, Ahmad I, Akbor MA, Rahman A, Manir MS, Cho KM. Unveiling chlorpyrifos mineralizing and tomato plant-growth activities of Enterobacter sp. strain HSTU-ASh6 using biochemical tests, field experiments, genomics, and in silico analyses. Front Microbiol. 2022; 13:1060554. doi:10.3389/fmicb.2022.1060554
- Hossain MS, Alam S, Wmburry L, Rahman A, Binduraz B, Roy S, Sultana MT, Ghosh S, Haque MA. Decoding genome, phylogenetic insights, plant-beneficial genetic repertoire, and in-silico pesticide biodegradation pathways of endophytic strain Serratia sp. HSTU-ABk35. J Biosci Public Health. 2026;2(1):30-54. doi:10.5455/JBPH.2025.20
- Barman DN, Haque MA, Islam SMA, Yun HD, Kim MK. Cloning and expression of ophB gene encoding organophosphorus hydrolase from endophytic Pseudomonas sp. BF1-3 degrades organophosphorus pesticide chlorpyrifos. Ecotoxicol Environ Saf. 2014; 108:135-141. doi: 10.1016/j.ecoenv.2014.06.023
- Chen S, Lai K, Li Y, Hu M, Zhang Y, Zeng Y. Biodegradation of chlorpyrifos by Bacillus subtilis strain isolated from contaminated soil and characterization of its degradation pathway. Int Biodeterior Biodegradation. 2015; 103:191-197. doi: 10.1016/j.ibiod.2015.04.012.
- Kumar S, Singh DK. Biodegradation of carbofuran by Bacillus cereus isolated from agricultural soil. J Environ Sci Health B. 2018;53(2):111-118. doi:10.1080/03601234.2017.1409336.
- Gangola S, Sharma A, Bhatt P. Biodegradation of carbofuran by Bacillus cereus isolated from agricultural soil and characterization of its degradation pathway. J Environ Chem Eng. 2021;9(2):105134. doi: 10.1016/j.jece.2021.105134.
- Abdelkader AA, Khalil MS, Mohamed MSM. Simultaneous biodegradation of λ‑cyhalothrin pesticide and Vicia faba growth promotion under greenhouse conditions. AMB Express. 2022; 12:44. doi:10.1186/s13568‑022‑01383‑0.
- Özdal M, Özdal ÖG, Algur ÖF, Kurbanoğlu EB. Biodegradation of α-endosulfan via hydrolysis pathway by Stenotrophomonas maltophilia OG2. 3 Biotech. 2017;7(2): Article 76. doi:10.1007/s13205-017-0765-y
- Parakhia MV, Tomar RS, Dalal H, Kothari VV, Rathod VM, Golakiya BA. Genome sequence analysis and identification of genes associated with pesticide degradation from Enterobacter cloacae strain MR2. Int J Curr Microbiol Appl Sci. 2019;8(1):2289‑2304. doi:10.20546/ijcmas.2019.801.240. I
- Lal R, Sharma P, Kumari K. Biochemistry of microbial degradation of hexachlorocyclohexane and prospects for bioremediation. Microbiol Mol Biol Rev. 2010;74(1):58‑80. doi:10.1128/MMBR.00014‑09.
- Pérez‑Pantoja D, De la Iglesia R, Pieper DH, González B. Cupriavidus necator JMP134 and the biodegradation of 2,4‑dichlorophenoxyacetic acid: metabolic pathways and ecological relevance. Appl Microbiol Biotechnol. 2012;94(4):1013‑1028. doi:10.1007/s00253‑011‑3723‑2. (Review on biodegradation pathways and ecology of C. necator JMP134)
- Cycoń M, Piotrowska‑Seget Z. Pyrethroid‑degrading microorganisms and their potential for the bioremediation of contaminated soils. Front Microbiol. 2016; 7:1463. doi:10.3389/fmicb.2016.01463.
- Grady EN, MacDonald J, Liu L, Richman A, Yuan Z‑C. Current knowledge and perspectives of Paenibacillus spp.: beneficial microbes in agriculture. Microbiol Mol Biol Rev. 2016;80(3):1‑43. doi:10.1128/MMBR.00036‑15.
- Cho DY, Jang MY, Lee HY, Jeong JB, Kim DH, Bang DY, Cho KM. Rhizospheric bacterial distribution influencing the accumulation of isoflavones, phenolics, flavonoids, and antioxidant activity in soybean roots within hydroponic system. Plants. 2025;14(14):2238. doi:10.3390/plants14142238
- DY, Haque MA, Lee HY, Jang MY, Jeong JB, Lee GY, et al. Amending metagenomic bacterial community in soybean-cultivated soils to enhance phytoestrogen in soybean roots by communicating with mixture of culturable rhizospheric bacteria. Plant Physiol Biochem. 2026; 232:111093. doi: 10.1016/j.plaphy.2026.111093.
- Roy MK, Roy S, Binduraz B, Afrin L, Haque MA. Pesticide-associated Health and Environmental Risks and the Potential of Biofertilizers in Sustainable Agriculture. J Biosci Public Health. 2025;1(3):16-27. https://doi.org/10.5455/JBPH.2025.12
- Abdullah-Al-Mamun M, Hossain MS, Debnath GC, Sultana S, Rahman A, Hasan Z, Haque MA. Unveiling lignocellulolytic trait of a goat omasum inhabitant Klebsiella variicola strain HSTU-AAM51 in light of biochemical and genome analyses. Braz J Microbiol. 2022;53(1):99-130. doi: 10.1007/s42770‑021‑00660‑7.
- Sharker B, Islam MA, Hossain MAA, Ahmad I, Al Mamun A, Ghosh S, Haque MA. Characterization of lignin and hemicellulose degrading bacteria isolated from cow rumen and forest soil: Unveiling a novel enzymatic model for rice straw deconstruction. Sci Total Environ. 2023; 904:166704. doi: 10.1016/j.scitotenv.2023.166704
- Haque AM, Hwang CE, Kim SC, Cho DY, Lee HY, Cho KM, Lee JH. Biodegradation of organophosphorus insecticides by two organophosphorus hydrolase genes (opdA and opdE) from isolated Leuconostoc mesenteroides WCP307 of kimchi origin. Process Biochem. 2020; 94:340-348. doi: 10.1016/j.procbio.2020.04.026
- Gopal A, Swamidason JTEY, Mariappan P, Bojan V. Microbial degradation of flubendiamide in different types of soils at tropical region using lactic acid bacteria formulation. Sci Rep. 2025;15(1):29271. doi:10.1038/s41598-025-08917-z
- Mohr S, Meinecke S, Feibicke M, Duquesne S, Frische T, Sahm R. Effects of a realistic pesticide spraying sequence for apple crop on stream communities in mesocosms: negligible or notable? Environ Sci Eur. 2023;35(1):35. doi:10.1186/s12302-023-00739-y
- Sharma A, Kumar V, Handa N, Bali S, Kaur R, Khanna K, Thukral A, Bhardwaj R. Potential of endophytic bacteria in heavy metal and pesticide detoxification. Plant Microbiome: Stress Response. 2018:307-336.
- Van der Plas M, Nederstigt TAP, Trimbos KB, Didaskalou EA, Vijver MG. Insights from a Long-Term Outdoor Mesocosm Study: eDNA Metabarcoding Reveals Exacerbated but Transient Impacts from a Nanoenabled Pesticide Formulation (Nano-TiO2-Coated Carbendazim) on Freshwater Microbial Communities. ACS ES T Water. 2025 Apr 22;5(5):2421-2431. doi: 10.1021/acsestwater.5c00014.
- Anusha YK, Sivakumar U, Manoranjitham SK, Senthilkumar M. Revolutionizing agriculture: Innovative bioinoculant formulation technologies for sustainability. Plant Sci Today. 2024; 11:5356. doi:10.14719/pst.5356
 access
access